
Exhibit 99.3

NDV - 01 12 - Month Phase 2 Data March 2026

©2026 Relmada - All rights reserved Disclosures 2 The Private Securities Litigation Reform Act of 1995 provides a safe harbor for forward - looking statements made by us or on our behalf . This press release contains statements which constitute “forward - looking statements” within the meaning of Section 27 A of the Securities Act of 1933 and Section 21 E of the Securities Exchange Act of 1934 . Any statement that is not historical in nature is a forward - looking statement and may be identified by the use of words and phrases such as “if”, “may”, “expects”, “anticipates”, “believes”, “will”, “will likely result”, “will continue”, “plans to”, “potential”, “promising”, and similar expressions . These statements are based on management’s current expectations and beliefs and are subject to a number of risks, uncertainties and assumptions that could cause actual results to differ materially from those described in the forward - looking statements, including potential for Phase 2 NDV - 01 data to continue to deliver positive results supporting further development, potential for clinical trials to deliver statistically and/or clinically significant evidence of efficacy and/or safety, failure of top - line results to accurately reflect the complete results of the trial, failure of planned or ongoing preclinical and clinical studies to demonstrate expected results, potential failure to secure FDA agreement on the regulatory path for sepranolone, and NDV - 01 , or that future sepranolone, or NDV - 01 clinical results will be acceptable to the FDA, failure to secure adequate sepranolone, or NDV - 01 drug supply, and the other risk factors described under the heading “Risk Factors” set forth in the Company’s reports filed with the SEC from time to time . No forward - looking statement can be guaranteed, and actual results may differ materially from those projected . Relmada undertakes no obligation to publicly update any forward - looking statement, whether as a result of new information, future events, or otherwise . Readers are cautioned that it is not possible to predict or identify all the risks, uncertainties and other factors that may affect future results and that the risks described herein should not be a complete list .
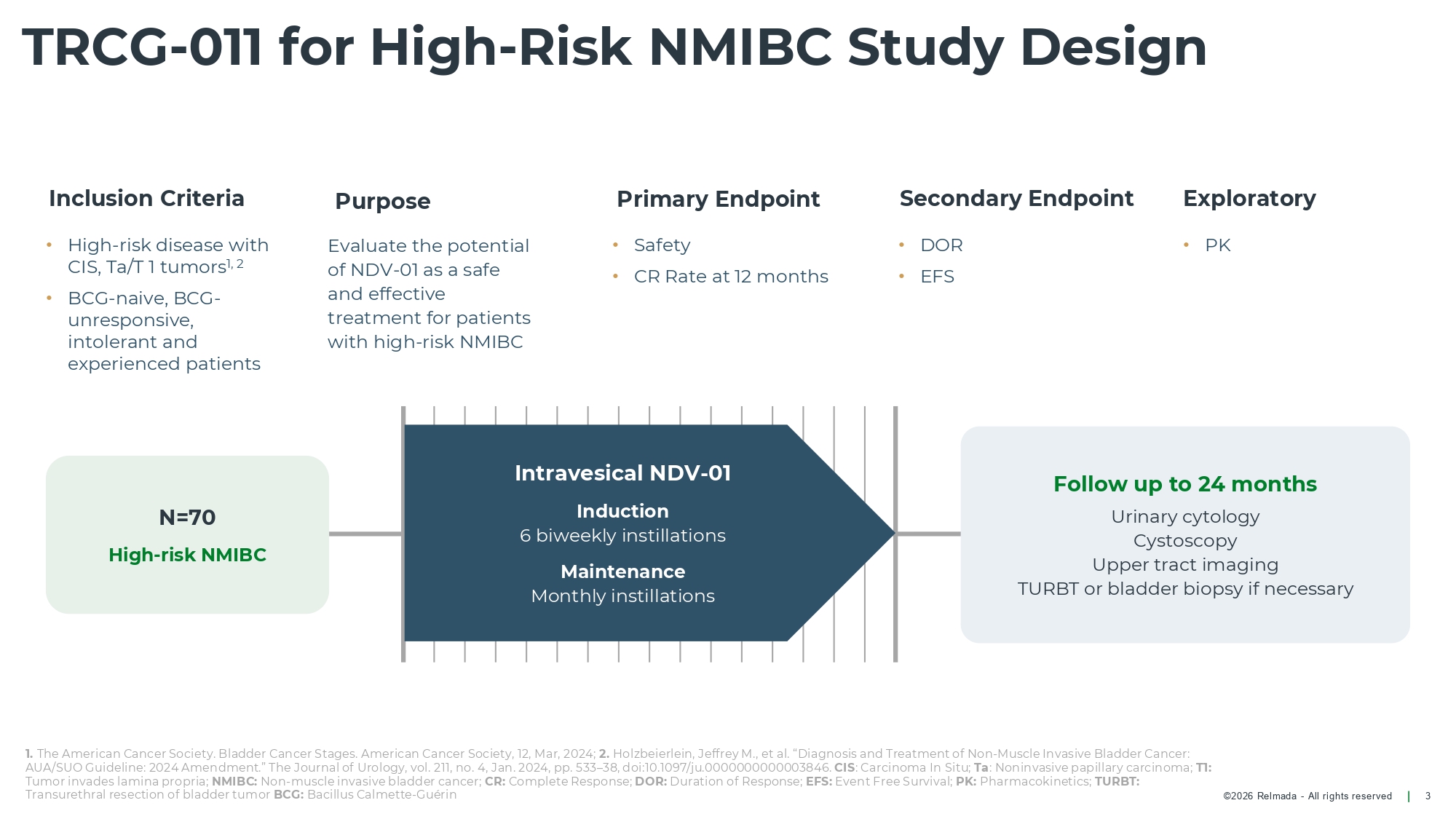
©2026 Relmada - All rights reserved Inclusion Criteria • High - risk disease with CIS, Ta/T 1 tumors 1, 2 • BCG - naive, BCG - unresponsive, intolerant and experienced patients Purpose Evaluate the potential of NDV - 01 as a safe and effective treatment for patients with high - risk NMIBC Primary Endpoint • Safety • CR Rate at 12 months Secondary Endpoint Exploratory • PK TRCG - 011 for High - Risk NMIBC Study Design 3 • DOR • EFS 1. The American Cancer Society. Bladder Cancer Stages. American Cancer Society, 12, Mar, 2024; 2. Holzbeierlein, Jeffrey M., et al. “Diagnosis and Treatment of Non - Muscle Invasive Bladder Cancer: AUA/SUO Guideline: 2024 Amendment.” The Journal of Urology, vol. 211, no. 4, Jan. 2024, pp. 533 – 38, doi:10.1097/ju.0000000000003846. CIS : Carcinoma In Situ; Ta : Noninvasive papillary carcinoma; T1: Tumor invades lamina propria; NMIBC: Non - muscle invasive bladder cancer; CR: Complete Response; DOR: Duration of Response; EFS: Event Free Survival; PK: Pharmacokinetics; TURBT: Transurethral resection of bladder tumor BCG: Bacillus Calmette - Guérin Follow up to 24 months Urinary cytology Cystoscopy Upper tract imaging TURBT or bladder biopsy if necessary N=70 High - risk NMIBC Intravesical NDV - 01 Induction 6 biweekly instillations Maintenance Monthly instillations
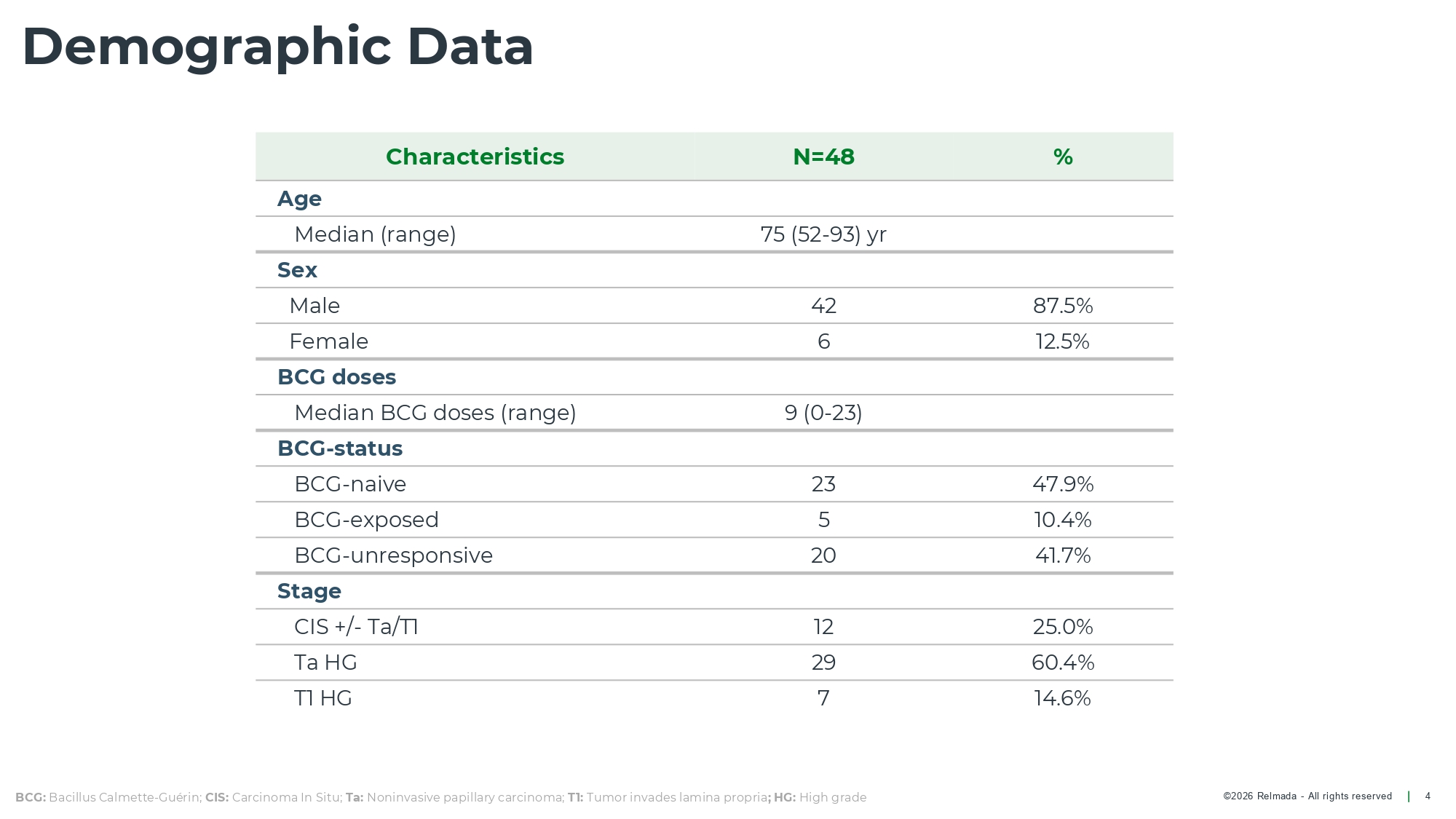
©2026 Relmada - All rights reserved Demographic Data 4 % N=48 Characteristics Age 75 (52 - 93) yr Median (range) Sex 87.5% 42 Male 12.5% 6 Female BCG doses 9 (0 - 23) Median BCG doses (range) BCG - status 47.9% 23 BCG - naive 10.4% 5 BCG - exposed 41.7% 20 BCG - unresponsive Stage 25.0% 12 CIS +/ - Ta/T1 60.4% 29 Ta HG 14.6% 7 T1 HG BCG: Bacillus Calmette - Guérin; CIS: Carcinoma In Situ; Ta: Noninvasive papillary carcinoma; T1: Tumor invades lamina propria ; HG: High grade
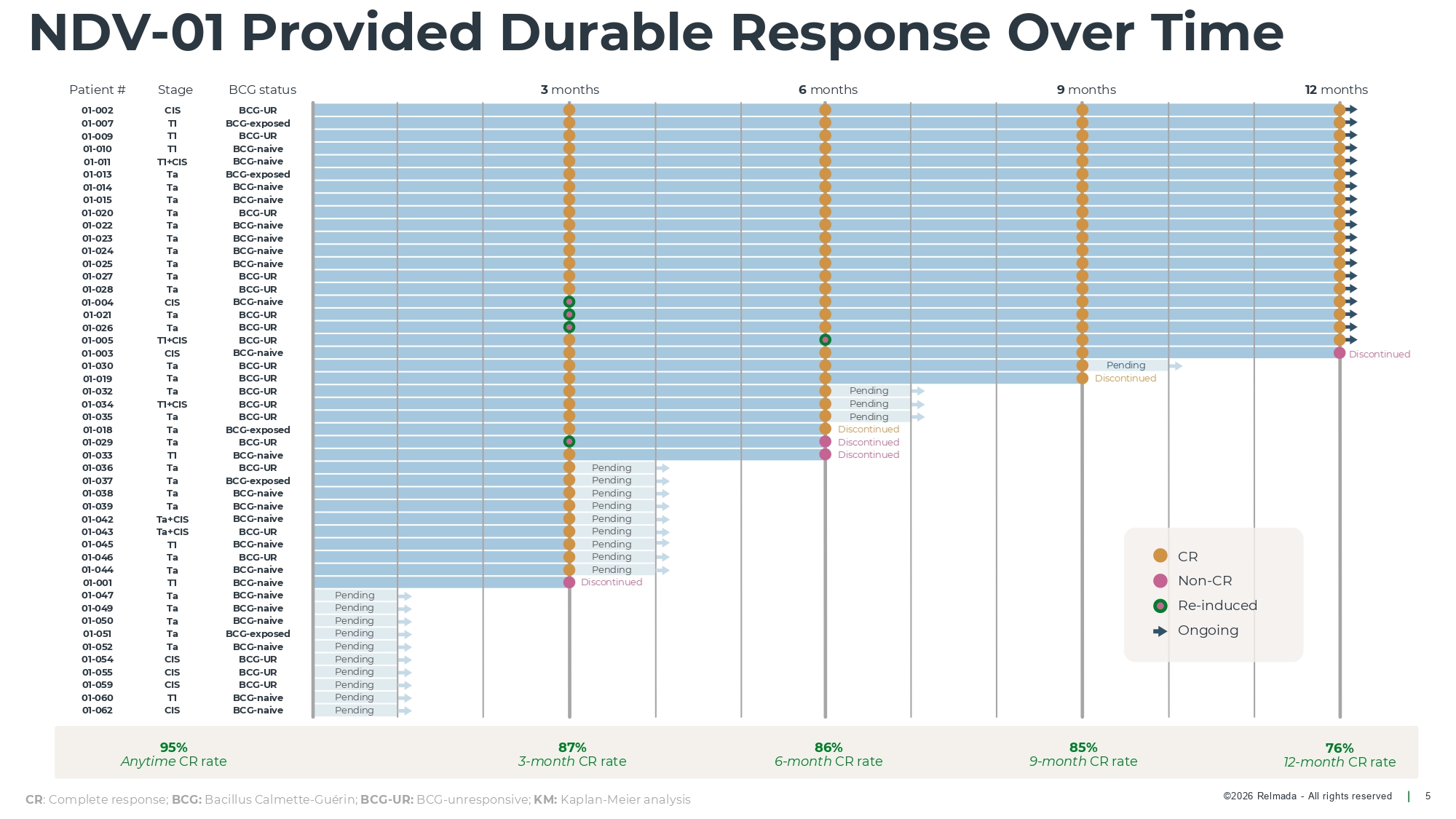
©2026 Relmada - All rights reserved 45. T1 BCG - naive Pending 46. Ta BCG - UR Pending 01 - 044 Ta BCG - naive Pending 01 - 001 T1 BCG - naive Discontinued 01 - 047 Ta BCG - naive Pending 01 - 049 Ta BCG - naive Pending 01 - 050 Ta BCG - naive Pending 01 - 051 Ta BCG - exposed Pending 01 - 052 Ta BCG - naive Pending 01 - 054 CIS BCG - UR Pending 01 - 055 CIS BCG - UR Pending 59. CIS BCG - UR Pending 60. T 1 BCG - naive Pending 01 - 062 CIS BCG - naive Pending 12 months NDV - 01 Provided Durable Response Over Time 5 Patient # Stage BCG status 3 months 6 months 9 months 01 - 002 CIS BCG - UR 01 - 007 T1 BCG - exposed 9. T1 BCG - UR 10. T1 BCG - naive 11. T1+CIS BCG - naive 13. Ta BCG - exposed 14. Ta BCG - naive 15. Ta BCG - naive 01 - 020 Ta BCG - UR 22. Ta BCG - naive 23. Ta BCG - naive 24. Ta BCG - naive 25. Ta BCG - naive 27. Ta BCG - UR 28. Ta BCG - UR 01 - 004 CIS BCG - naive 01 - 021 Ta BCG - UR 01 - 026 Ta BCG - UR 01 - 005 T1+CIS BCG - UR 01 - 003 CIS BCG - naive 01 - 030 Ta BCG - UR Pending 01 - 019 Ta BCG - UR Discontinued 01 - 032 Ta BCG - UR Pending 34. T1+CIS BCG - UR Pending 35. Ta BCG - UR Pending 01 - 018 Ta BCG - exposed Discontinued 01 - 029 Ta BCG - UR Discontinued 01 - 033 T1 BCG - naive Discontinued 36. Ta BCG - UR Pending 37. Ta BCG - exposed Pending 38. Ta BCG - naive Pending 39. Ta BCG - naive Pending 42. Ta+CIS BCG - naive Pending 43. Ta+CIS BCG - UR Pending Discontinued 95% Anytime CR rate 87% 3 - month CR rate 86% 6 - month CR rate 85% 9 - month CR rate CR : Complete response; BCG: Bacillus Calmette - Guérin; BCG - UR: BCG - unresponsive; KM: Kaplan - Meier analysis 76% 12 - month CR rate CR Non - CR Re - induced Ongoing
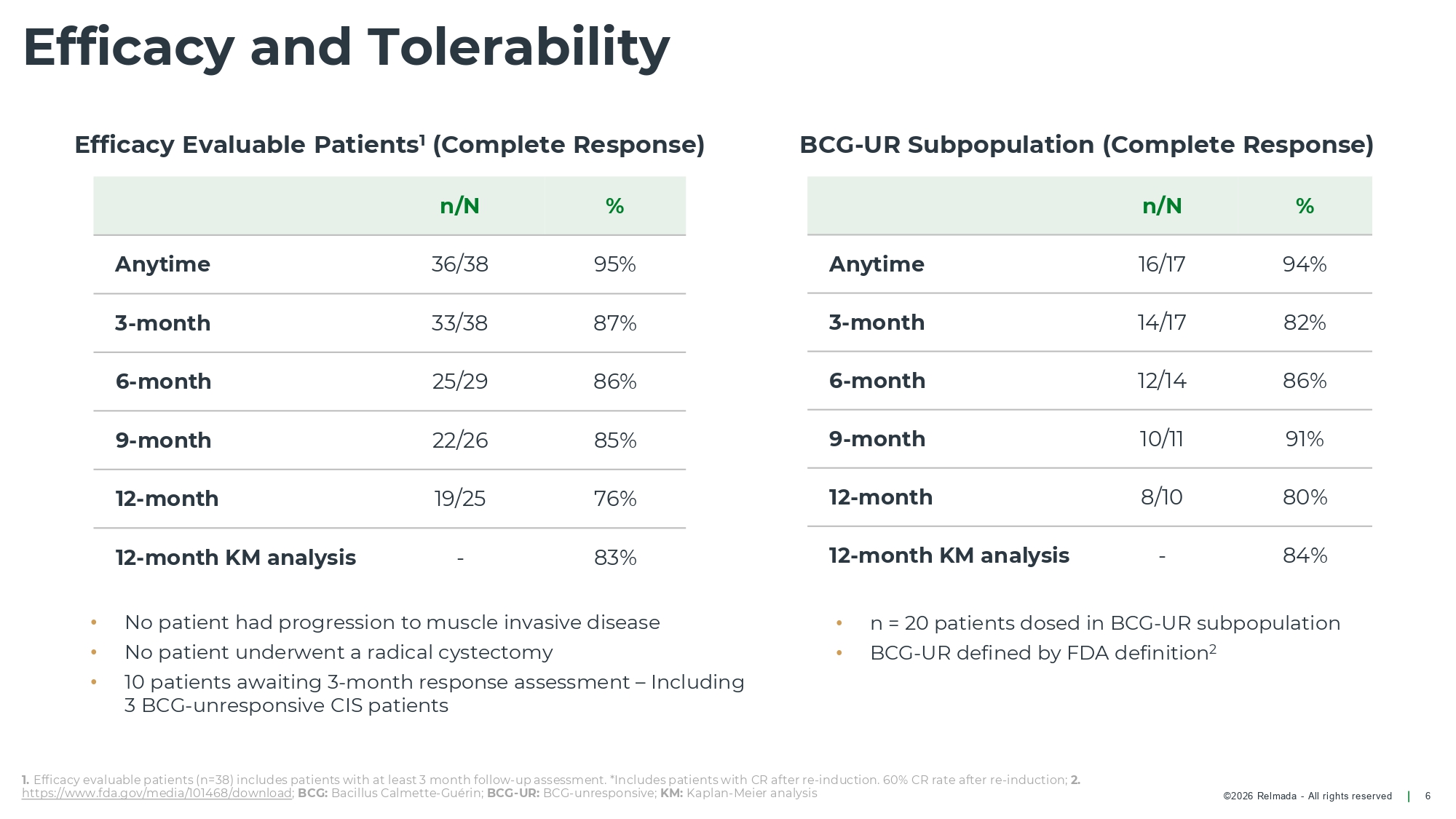
©2026 Relmada - All rights reserved Efficacy and Tolerability 6 % n/N 95% 36/38 Anytime 87% 33/38 3 - month 86% 25/29 6 - month 85% 22/26 9 - month 76% 19/25 12 - month 83% - 12 - month KM analysis • No patient had progression to muscle invasive disease • No patient underwent a radical cystectomy • 10 patients awaiting 3 - month response assessment – Including 3 BCG - unresponsive CIS patients 1. Efficacy evaluable patients (n=38) includes patients with at least 3 month follow - up assessment. *Includes patients with CR after re - induction. 60% CR rate after re - induction; 2. https://www.fda.gov/media/101468/download ; BCG: Bacillus Calmette - Guérin; BCG - UR: BCG - unresponsive; KM: Kaplan - Meier analysis % n/N 94% 16/17 Anytime 82% 14/17 3 - month 86% 12/14 6 - month 91% 10/11 9 - month 80% 8/10 12 - month 84% - 12 - month KM analysis • n = 20 patients dosed in BCG - UR subpopulation • BCG - UR defined by FDA definition 2 BCG - UR Subpopulation (Complete Response) Efficacy Evaluable Patients 1 (Complete Response)

©2026 Relmada - All rights reserved Treatment - Related AE and Tolerability 7 No patient had ≥ Grade 3 TRAE No patients discontinued treatment due to AEs Of the 48 patients who received ≥ 1 dose of NDV - 01, 30 (63%) had a TRAE • 54% transient uncomfortable urination (dysuria) • 8% asymptomatic positive urine culture • 8% hematuria TRAE: Treatment - related adverse events; AE: Adverse events
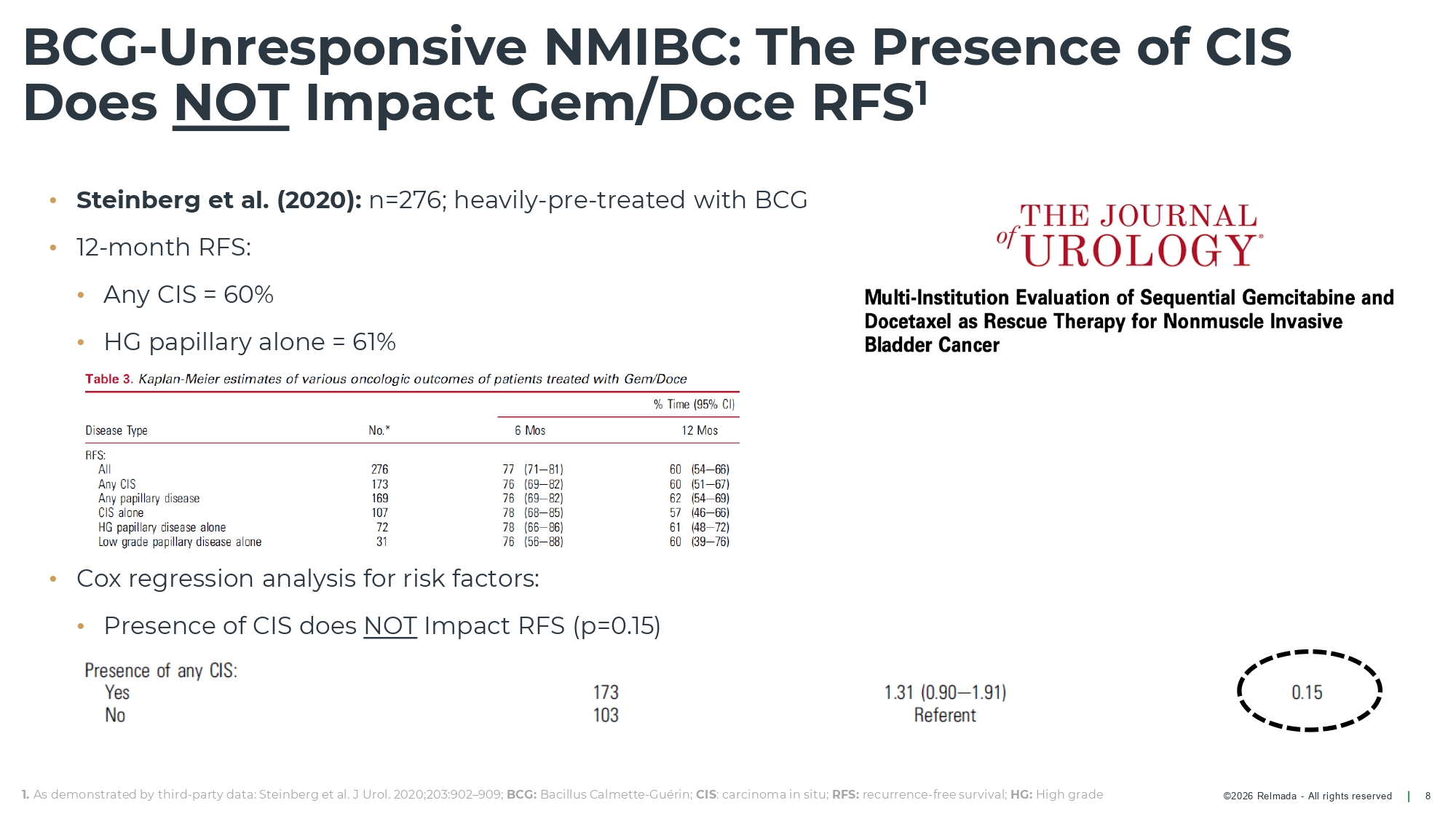
©2026 Relmada - All rights reserved • Steinberg et al. (2020): n=276; heavily - pre - treated with BCG • 12 - month RFS: • Any CIS = 60% • HG papillary alone = 61% • Cox regression analysis for risk factors: • Presence of CIS does NOT Impact RFS (p=0.15) BCG - Unresponsive NMIBC: The Presence of CIS Does NOT Impact Gem/Doce RFS 1 8 1. As demonstrated by third - party data: Steinberg et al. J Urol. 2020;203:902 – 909; BCG: Bacillus Calmette - Guérin; CIS : carcinoma in situ; RFS: recurrence - free survival; HG: High grade


©2026 Relmada - All rights reserved Two Independent NDV - 01 Approval Pathways Provide Significant Market Opportunity 10 1. Based on Internal estimates. 2. Grabe - Heyne et al. Front Oncol. 2023. 3. FDA approval summaries; company disclosures; published clinical trial data. NMIBC: Non - muscle invasive bladder cancer; BCG: Bacillus Calmette - Guérin (BCG); TURBT: Transurethral Resection of Bladder Tumor; CIS : carcinoma in situ; 1L: first - line; 2L: second - line; CR : Complete Response; ~75k patients/annually in US 1 – with ~35% 2 of intermediate - risk patients receiving adjuvant therapy post - TURBT Registrational Pathway 1 Open label randomized controlled trial in intermediate - risk NMIBC – adjuvant therapy following TURBT (NDV - 01 vs. observation) ~5k patients/annually in US 1 – based on 12 - month CR rates of 19% - 46% 3 for 1L BCG - unresponsive therapies Registrational Pathway 2 Single - arm trial in 2L BCG - unresponsive NMIBC with CIS who are refractory to approved or developmental therapies R ecurrent / E ndovesical / S urgery - sparing / C ombination therapy for / U rothelial cancer / E ffectiveness
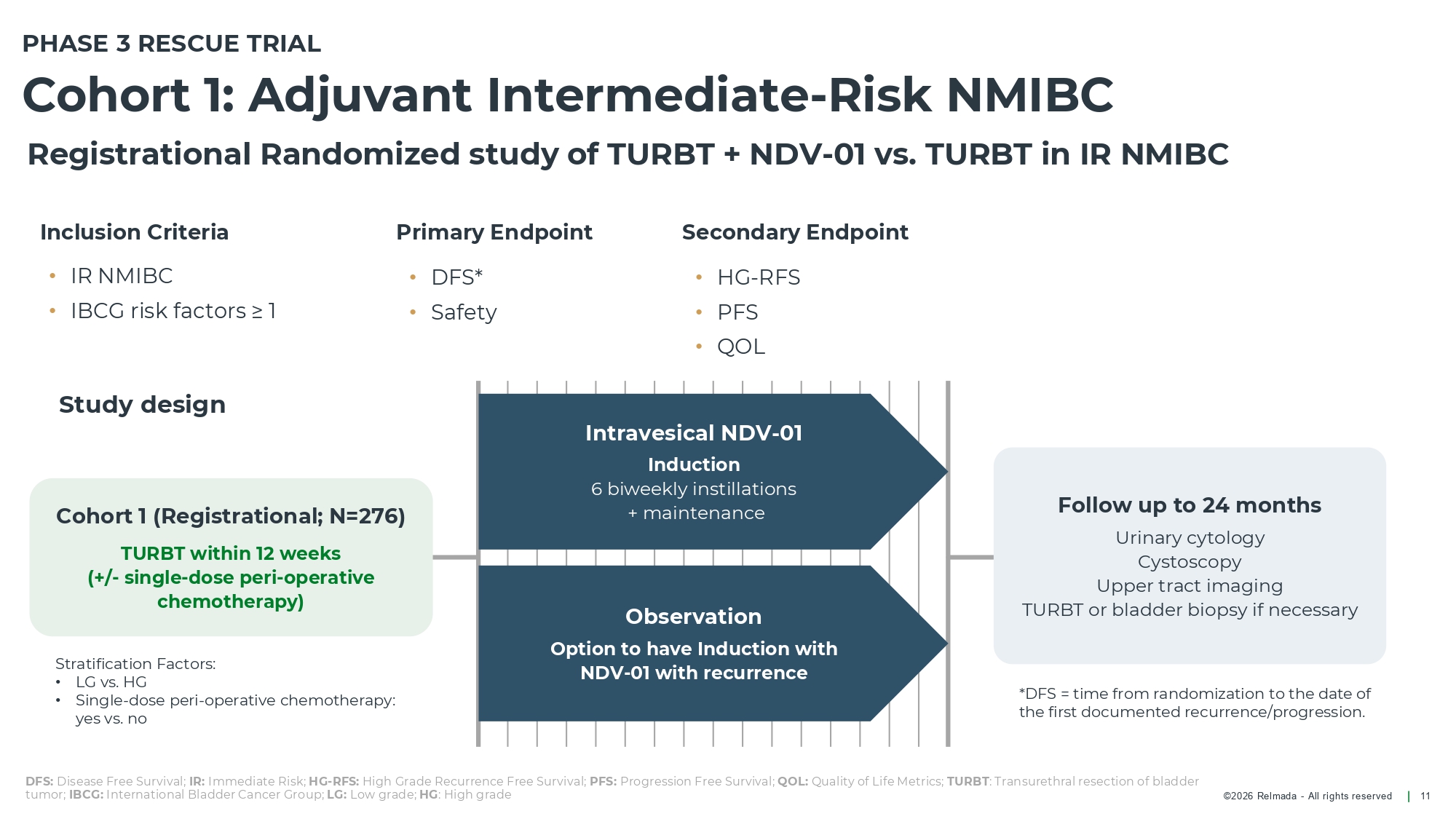
©2026 Relmada - All rights reserved Cohort 1: Adjuvant Intermediate - Risk NMIBC 11 Registrational Randomized study of TURBT + NDV - 01 vs. TURBT in IR NMIBC Inclusion Criteria • IR NMIBC • IBCG risk factors ≥ 1 Primary Endpoint • DFS* • Safety Intravesical NDV - 01 Induction 6 biweekly instillations + maintenance Observation Option to have Induction with NDV - 01 with recurrence Follow up to 24 months Urinary cytology Cystoscopy Upper tract imaging TURBT or bladder biopsy if necessary DFS: Disease Free Survival; IR: Immediate Risk; HG - RFS: High Grade Recurrence Free Survival; PFS: Progression Free Survival; QOL: Quality of Life Metrics; TURBT : Transurethral resection of bladder tumor; IBCG: International Bladder Cancer Group; LG: Low grade; HG : High grade Cohort 1 (Registrational; N=276) TURBT within 12 weeks (+/ - single - dose peri - operative chemotherapy) Study design *DFS = time from randomization to the date of the first documented recurrence/progression. PHASE 3 RESCUE TRIAL Stratification Factors: • LG vs. HG • Single - dose peri - operative chemotherapy: yes vs. no Secondary Endpoint • HG - RFS • PFS • QOL

©2026 Relmada - All rights reserved Cohort 2A: 2L BCG - Unresponsive NMIBC 12 Open - label, single - arm study to evaluate safety and efficacy of NDV - 01 in BCG - UR refractory to first - line therapy Study design PHASE 3 RESCUE TRIAL Inclusion Criteria Purpose • HR BCG - UR with CIS refractory to first - line therapy • Safety and efficacy of NDV - 01 in patients with HR BCG - UR with CIS Secondary Endpoint Primary Endpoint Other • CR anytime • Safety • DOR • PFS • RFS amongst responders • PK 1. BCG - Unresponsive patients with CIS +/ - Ta/T1 disease. Phase 3 Cohort 2A is a registrational cohort intended for regulatory approval. 2. BCG - Unresponsive patients with high - grade Ta/T1 disease. Phase 2 Cohort 2B is an exploratory cohort and not intended for regulatory approval. HR: High risk; CIS: Carcinoma In Situ; CR: Complete Response; DOR: Duration of Response; RFS: Recurrence Free Survival; PFS: Progression Free Survival; PK: Pharmacokinetics; TURBT: Transurethral resection of bladder tumor; BCG: Bacillus Calmette - Guérin BCG - UR: BCG - unresponsive Intravesical NDV - 01 Induction 6 biweekly instillations Maintenance Monthly instillations Follow up to 24 months Urinary cytology Cystoscopy TURBT or bladder biopsy if necessary Cohort 2A 1 (Registrational; N=87) HR BCG - UR NMIBC with CIS refractory to 1 st line therapy