
Exhibit 99.2

Exhibit 99.2 C O R P O R A T E O V E R V I E W Unlocking Life Changing Therapies March 2026

Disclosures The Private Securities Litigation Reform Act of 1995 provides a safe harbor for forward - looking statements made by us or on our behalf. This press release contains statements which constitute “forward - looking statements” within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Any statement that is not historical in nature is a forward - looking statement and may be identified by the use of words and phrases such as “if”, “may”, “expects”, “anticipates”, “believes”, “will”, “will likely result”, “will continue”, “plans to”, “potential”, “promising”, and similar expressions. These statements are based on management’s current expectations and beliefs and are subject to a number of risks, uncertainties and assumptions that could cause actual results to differ materially from those described in the forward - looking statements, including potential for Phase 2 NDV - 01 data to continue to deliver positive results supporting further development, potential for clinical trials to deliver statistically and/or clinically significant evidence of efficacy and/or safety, failure of interim or top - line results to accurately reflect the complete results of the trial, failure of planned or ongoing preclinical and clinical studies to demonstrate expected results, potential failure to secure FDA agreement on the regulatory path for sepranolone, and NDV - 01, or that future sepranolone, or NDV - 01 clinical results will be acceptable to the FDA, failure to secure adequate sepranolone, or NDV - 01 drug supply, and the other risk factors described under the heading “Risk Factors” set forth in the Company’s reports filed with the SEC from time to time. No forward - looking statement can be guaranteed, and actual results may differ materially from those projected. Relmada undertakes no obligation to publicly update any forward - looking statement, whether as a result of new information, future events, or otherwise. Readers are cautioned that it is not possible to predict or identify all the risks, uncertainties and other factors that may affect future results and that the risks described herein should not be a complete list. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any state or jurisdiction in which such offer, solicitation, or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. ©2026 Relmada - All rights reserved 2

Investment Thesis Innovative pipeline of potential high - value assets , led by NDV - 01 for non - muscle invasive bladder cancer (NMIBC) NDV - 01, a late - stage sustained - release Gem/Doce with attractive commercial profile and well - defined regulatory pathway Improvement vs. conventional Gem/Doce , positioning NDV - 01 as a next - generation standard - of - care driven by ease and speed of administration, extended tumor exposure and physician familiarity Proven efficacy of conventional Gem/Doce supported by positive clinical response and tolerability profile for NDV - 01 reduce mechanistic and regulatory risk Experienced leadership team supported by leading urology KOLs with direct NMIBC trial and practice experience ©2026 Relmada - All rights reserved 3
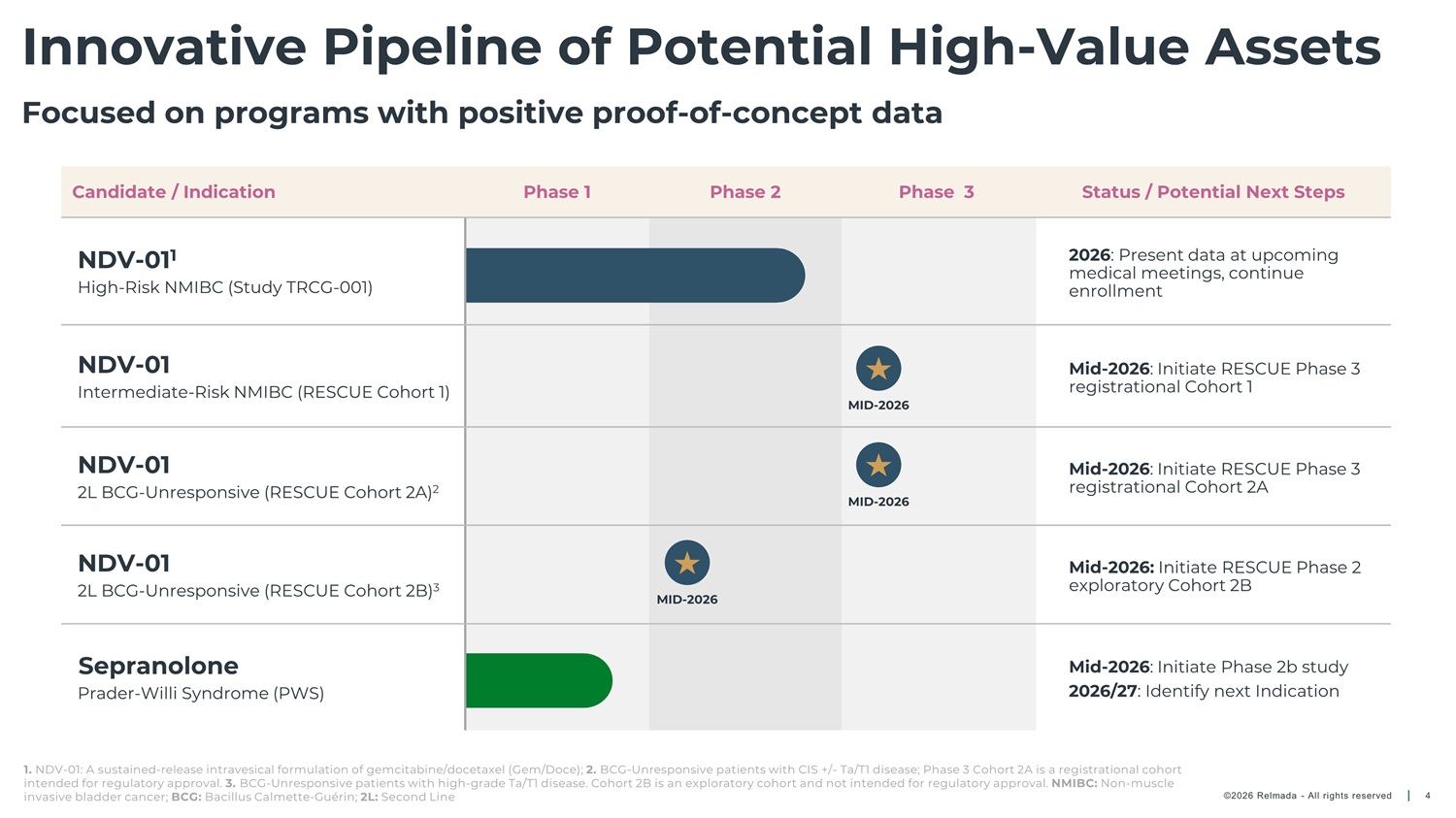
Innovative Pipeline of Potential High - Value Assets Focused on programs with positive proof - of - concept data Candidate / Indication Phase 1 Phase 2 Phase 3 Status / Potential Next Steps NDV - 01 1 High - Risk NMIBC (Study TRCG - 001) 2026 : Present data at upcoming medical meetings, continue enrollment NDV - 01 Mid - 2026 : Initiate RESCUE Phase 3 registrational Cohort 1 Intermediate - Risk NMIBC (RESCUE Cohort 1) MID - 2026 NDV - 01 2L BCG - Unresponsive (RESCUE Cohort 2A) 2 Mid - 2026 : Initiate RESCUE Phase 3 registrational Cohort 2A MID - 2026 NDV - 01 2L BCG - Unresponsive (RESCUE Cohort 2B) 3 Mid - 2026: Initiate RESCUE Phase 2 exploratory Cohort 2B MID - 2026 Sepranolone Prader - Willi Syndrome (PWS) Mid - 2026 : Initiate Phase 2b study 2026/27 : Identify next Indication 1. NDV - 01: A sustained - release intravesical formulation of gemcitabine/docetaxel (Gem/Doce); 2. BCG - Unresponsive patients with CIS +/ - Ta/T1 disease; Phase 3 Cohort 2A is a registrational cohort intended for regulatory approval. 3. BCG - Unresponsive patients with high - grade Ta/T1 disease. Cohort 2B is an exploratory cohort and not intended for regulatory approval. NMIBC: Non - muscle invasive bladder cancer; BCG: Bacillus Calmette - Guérin; 2L: Second Line ©2026 Relmada - All rights reserved 4

NDV - 01 A sustained - release intravesical formulation of gemcitabine/docetaxel (Gem/Doce) for patients with NMIBC, with positive Phase 2a data 1 1. Relmada press release March 9, 2025 NMIBC: Non - muscle invasive bladder cancer. The graphic is for artistic purposes only, not a factual representation ©2026 Relmada - All rights reserved 5
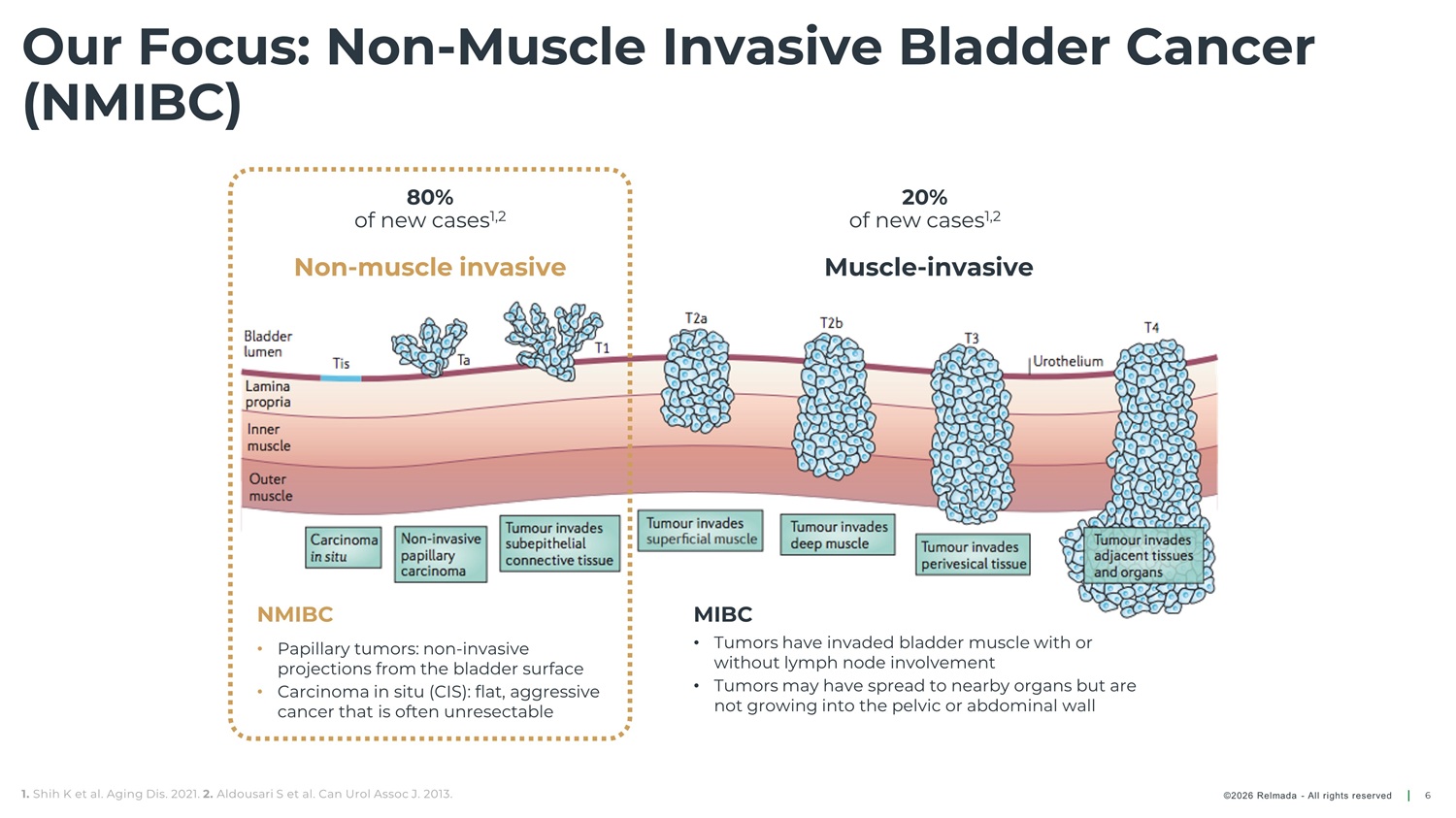
Our Focus: Non - Muscle Invasive Bladder Cancer (NMIBC) 80% 20% of new cases 1,2 of new cases 1,2 Non - muscle invasive Muscle - invasive NMIBC MIBC • Tumors have invaded bladder muscle with or • Papillary tumors: non - invasive without lymph node involvement projections from the bladder surface • Tumors may have spread to nearby organs but are • Carcinoma in situ (CIS): flat, aggressive not growing into the pelvic or abdominal wall cancer that is often unresectable 1. Shih K et al. Aging Dis. 2021. 2. Aldousari S et al. Can Urol Assoc J. 2013. ©2026 Relmada - All rights reserved 6
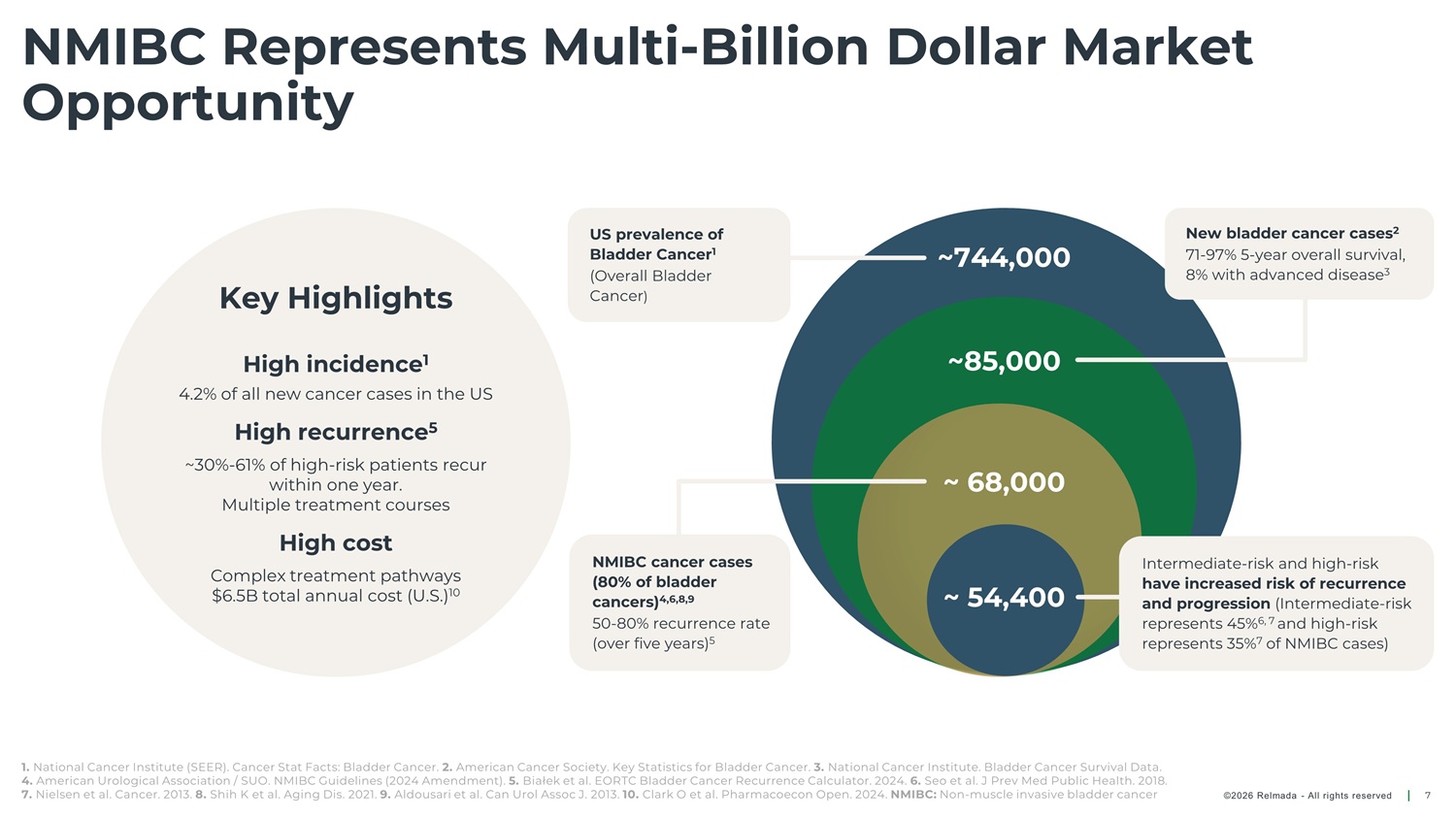
NMIBC Represents Multi - Billion Dollar Market Opportunity US prevalence of Bladder Cancer 1 (Overall Bladder Cancer ) New bladder cancer cases 2 71 - 97% 5 - year overall survival, 8% with advanced disease 3 ~744,000 ~85,000 Key Highlights High incidence 1 4.2% of all new cancer cases in the US High recurrence 5 ~30% - 61% of high - risk patients recur within one year. ~ 68,000 ~ 54,400 Multiple treatment courses High cost NMIBC cancer cases (80% of bladder cancers) 4,6,8,9 50 - 80% recurrence rate (over five years) 5 Intermediate - risk and high - risk have increased risk of recurrence and progression (Intermediate - risk represents 45% 6, 7 and high - risk represents 35% 7 of NMIBC cases) Complex treatment pathways $6.5B total annual cost (U.S.) 10 1. National Cancer Institute (SEER). Cancer Stat Facts: Bladder Cancer. 2. American Cancer Society. Key Statistics for Bladder Cancer. 3. National Cancer Institute. Bladder Cancer Survival Data. 4. American Urological Association / SUO. NMIBC Guidelines (2024 Amendment). 5. Białek et al. EORTC Bladder Cancer Recurrence Calculator. 2024. 6. Seo et al. J Prev Med Public Health. 2018. 7. Nielsen et al. Cancer. 2013. 8. Shih K et al. Aging Dis. 2021. 9. Aldousari et al. Can Urol Assoc J. 2013. 10. Clark O et al. Pharmacoecon Open. 2024. NMIBC: Non - muscle invasive bladder cancer ©2026 Relmada - All rights reserved 7
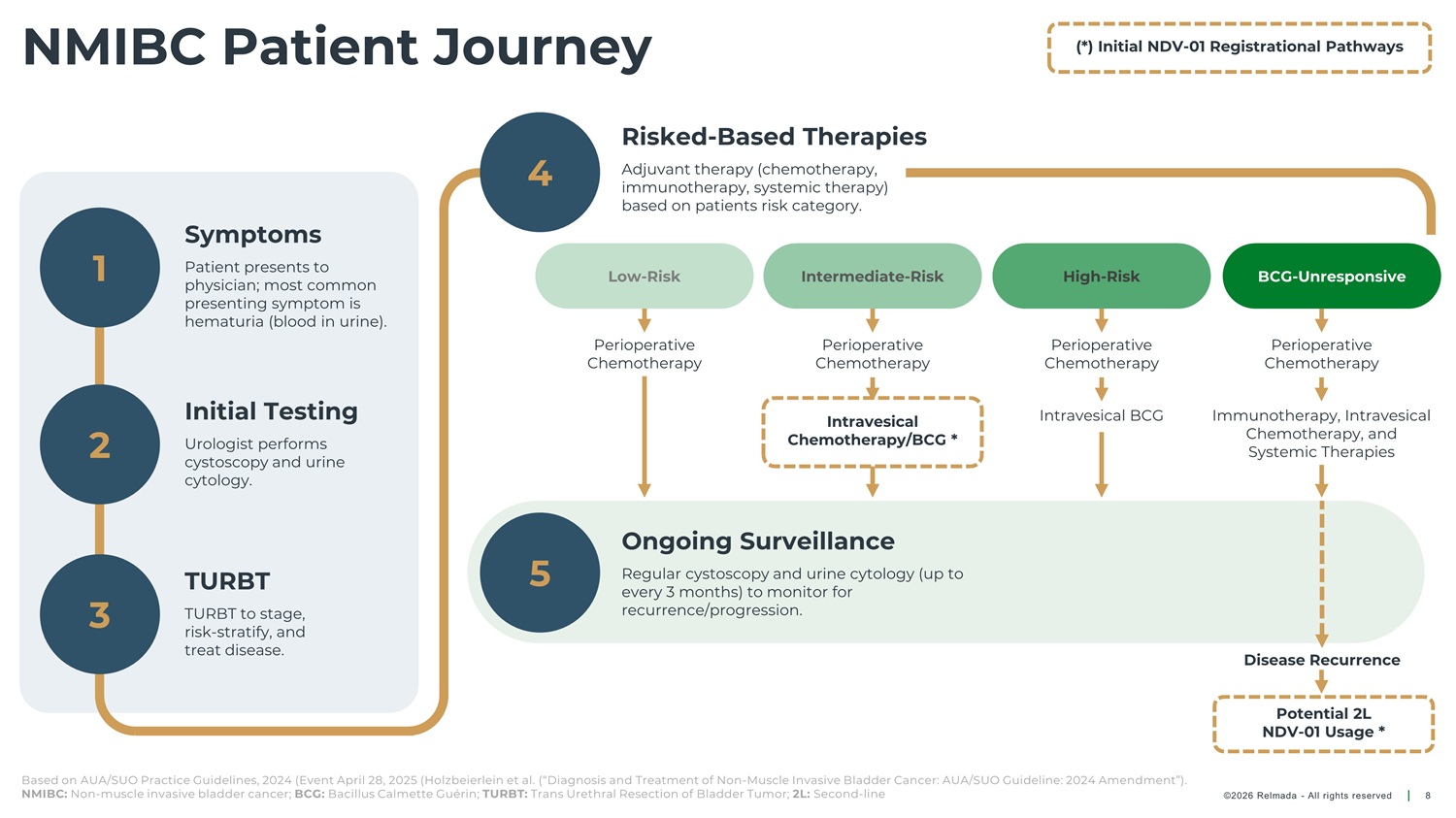
NMIBC Patient Journey (*) Initial NDV - 01 Registrational Pathways Risked - Based Therapies 4 Adjuvant therapy (chemotherapy, immunotherapy, systemic therapy) based on patients risk category. Symptoms 1 Patient presents to Low - Risk Intermediate - Risk High - Risk BCG - Unresponsive physician; most common presenting symptom is hematuria (blood in urine). Perioperative Chemotherapy Perioperative Chemotherapy Perioperative Chemotherapy Perioperative Chemotherapy Initial Testing Intravesical BCG Immunotherapy, Intravesical Chemotherapy, and Intravesical 2 3 Chemotherapy/BCG * Urologist performs cystoscopy and urine cytology. Systemic Therapies Ongoing Surveillance 5 TURBT Regular cystoscopy and urine cytology (up to every 3 months) to monitor for recurrence/progression. TURBT to stage, risk - stratify, and treat disease. Disease Recurrence Potential 2L NDV - 01 Usage * Based on AUA/SUO Practice Guidelines, 2024 (Event April 28, 2025 (Holzbeierlein et al. (“Diagnosis and Treatment of Non - Muscle Invasive Bladder Cancer: AUA/SUO Guideline: 2024 Amendment”). NMIBC: Non - muscle invasive bladder cancer; BCG: Bacillus Calmette Guérin; TURBT: Trans Urethral Resection of Bladder Tumor; 2L: Second - line ©2026 Relmada - All rights reserved 8

Overview of NMIBC Treatment Landscape Approved and emerging treatments TURBT Surgery Intravesical Chemotherapy Gene Therapy/ Immunotherapy Systemic Therapy Complications (>15%) 1 Emerging dataset Risk of recurrence (50 - 80%) 2 Supply issues Risk of recurrence OR procedure under anesthesia Conventional Chemotherapies : mitomycin, gemcitabine, Gem/Doce Risk of immune - mediated or systemic side effects Complex handling requirements Patient burden Sustained - Release : NDV - 01 (Gem/Doce), INLEXZO (gemcitabine), ZUSDURI (mitomycin) KEYTRUDA® (anti - PD1), Sasanlimab (anti - PD1), TYRA - 300 (oral FGFR3) BCG, Adstiladrin®, Anktiva®, Cretostimogene, TARA - 002, EG - 70, TAR - 210 (FGFR inhibitor) Relmada internal market research, 2025. 1. Pycha A et al. Urology. 2003. 2. Białek, Ł. (2024, August 1). EORTC Bladder Cancer Recurrence and Progression Calculator . Omni Calculator. NMIBC: Non - muscle invasive bladder cancer; TURBT: Transurethral resection of bladder tumor; BCG: Bacillus Calmette - Guérin ©2026 Relmada - All rights reserved 9
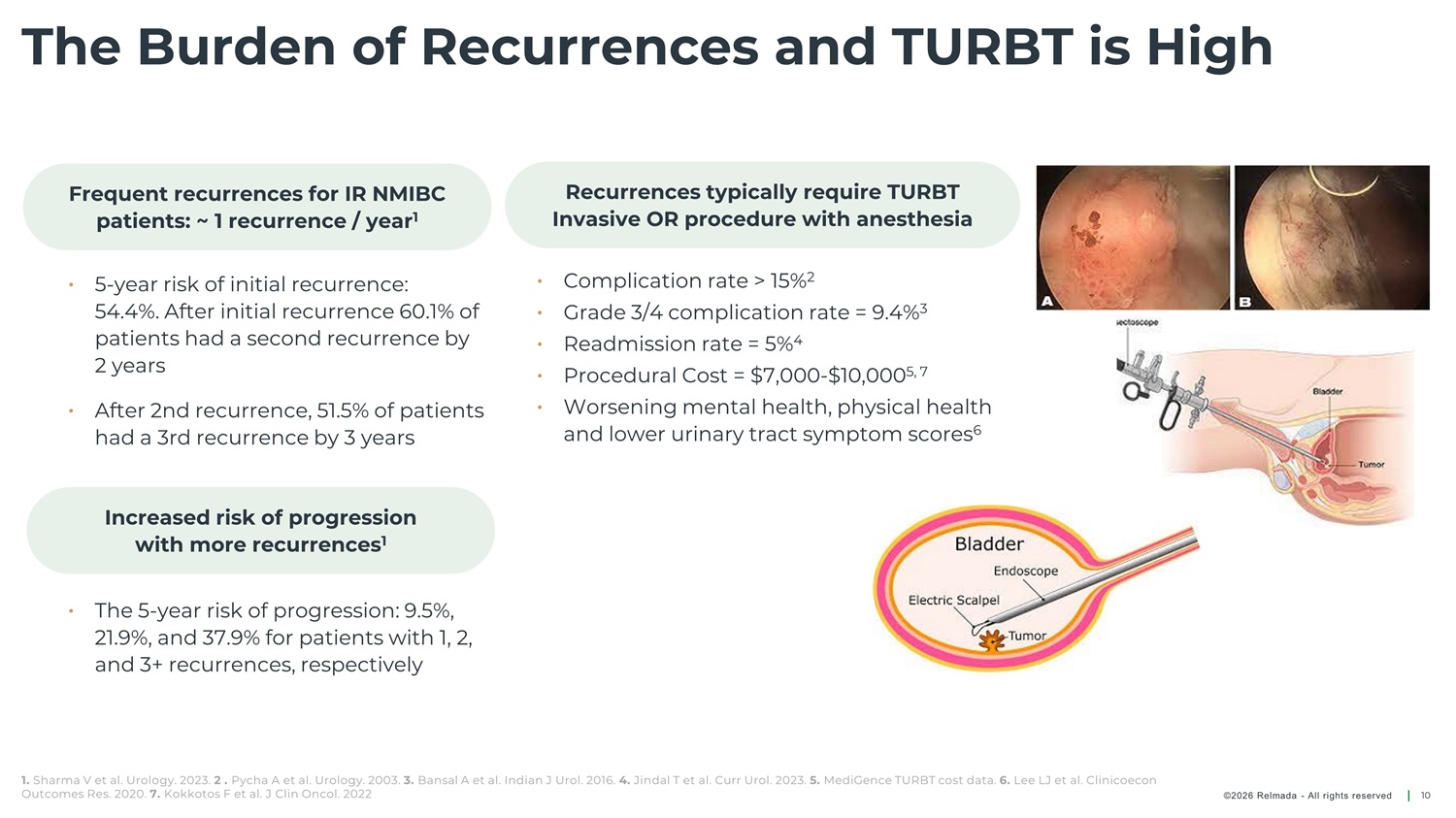
The Burden of Recurrences and TURBT is High Frequent recurrences for IR NMIBC patients: ~ 1 recurrence / year 1 Recurrences typically require TURBT Invasive OR procedure with anesthesia • Complication rate > 15% 2 • 5 - year risk of initial recurrence: 54.4%. After initial recurrence 60.1% of patients had a second recurrence by 2 years • Grade 3/4 complication rate = 9.4% 3 • Readmission rate = 5% 4 • Procedural Cost = $7,000 - $10,000 5, 7 • Worsening mental health, physical health and lower urinary tract symptom scores 6 • After 2nd recurrence, 51.5% of patients had a 3rd recurrence by 3 years Increased risk of progression with more recurrences 1 • The 5 - year risk of progression: 9.5%, 21.9%, and 37.9% for patients with 1, 2, and 3+ recurrences, respectively 1. Sharma V et al. Urology. 2023. 2 . Pycha A et al. Urology. 2003. 3. Bansal A et al. Indian J Urol. 2016. 4. Jindal T et al. Curr Urol. 2023. 5. MediGence TURBT cost data. 6. Lee LJ et al. Clinicoecon Outcomes Res. 2020. 7. Kokkotos F et al. J Clin Oncol. 2022 ©2026 Relmada - All rights reserved 10
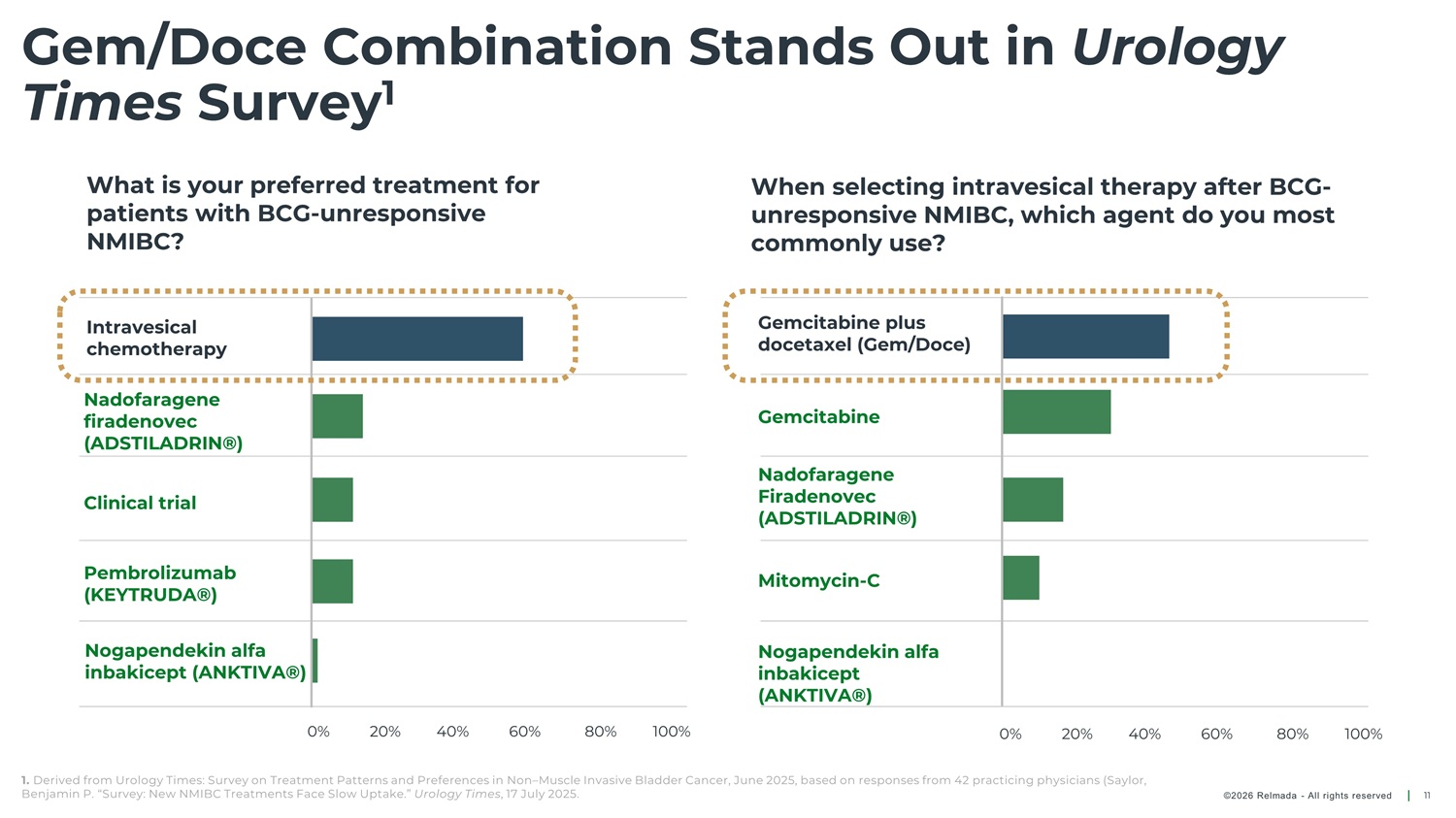
Gem/Doce Combination Stands Out in Urology Times Survey 1 What is your preferred treatment for patients with BCG - unresponsive NMIBC? When selecting intravesical therapy after BCG - unresponsive NMIBC, which agent do you most commonly use? Gemcitabine plus docetaxel (Gem/Doce) Intravesical chemotherapy Nadofaragene firadenovec (ADSTILADRIN®) Gemcitabine Nadofaragene Firadenovec (ADSTILADRIN®) Clinical trial Pembrolizumab (KEYTRUDA®) Mitomycin - C Nogapendekin alfa inbakicept (ANKTIVA®) Nogapendekin alfa inbakicept (ANKTIVA®) 0% 20% 40% 60% 80% 100% 0% 20% 40% 60% 80% 100% 1. Derived from Urology Times: Survey on Treatment Patterns and Preferences in Non – Muscle Invasive Bladder Cancer, June 2025, based on responses from 42 practicing physicians (Saylor, Benjamin P. “Survey: New NMIBC Treatments Face Slow Uptake.” Urology Times , 17 July 2025. ©2026 Relmada - All rights reserved 11

Significant Issues with Conventional Gem/Doce Intravesical Therapy for NMIBC Preparation by specialized pharmacy First administration 4 - hour total procedure time vs. <5 minutes for NDV - 01 Requires specialized pharmacy preparation vs. NDV - 01 comes ready for use in two pre - filled plastic syringes ~60 - 120 min dwell time 30 min break vs. NDV - 01 which provides the opportunity for community urologist to deliver Gem/Doce Utilization concentrated in academic setting Second administration ~90 - 120 min dwell time vs. NDV - 01 which provides sustained release of Gem/Doce for up to 10 days Limited drug exposure and dwell time NMIBC : Non - muscle invasive bladder cancer; Gem/Doce : Gemcitabine plus Docetaxel 12 ©2026 Relmada - All rights reserved
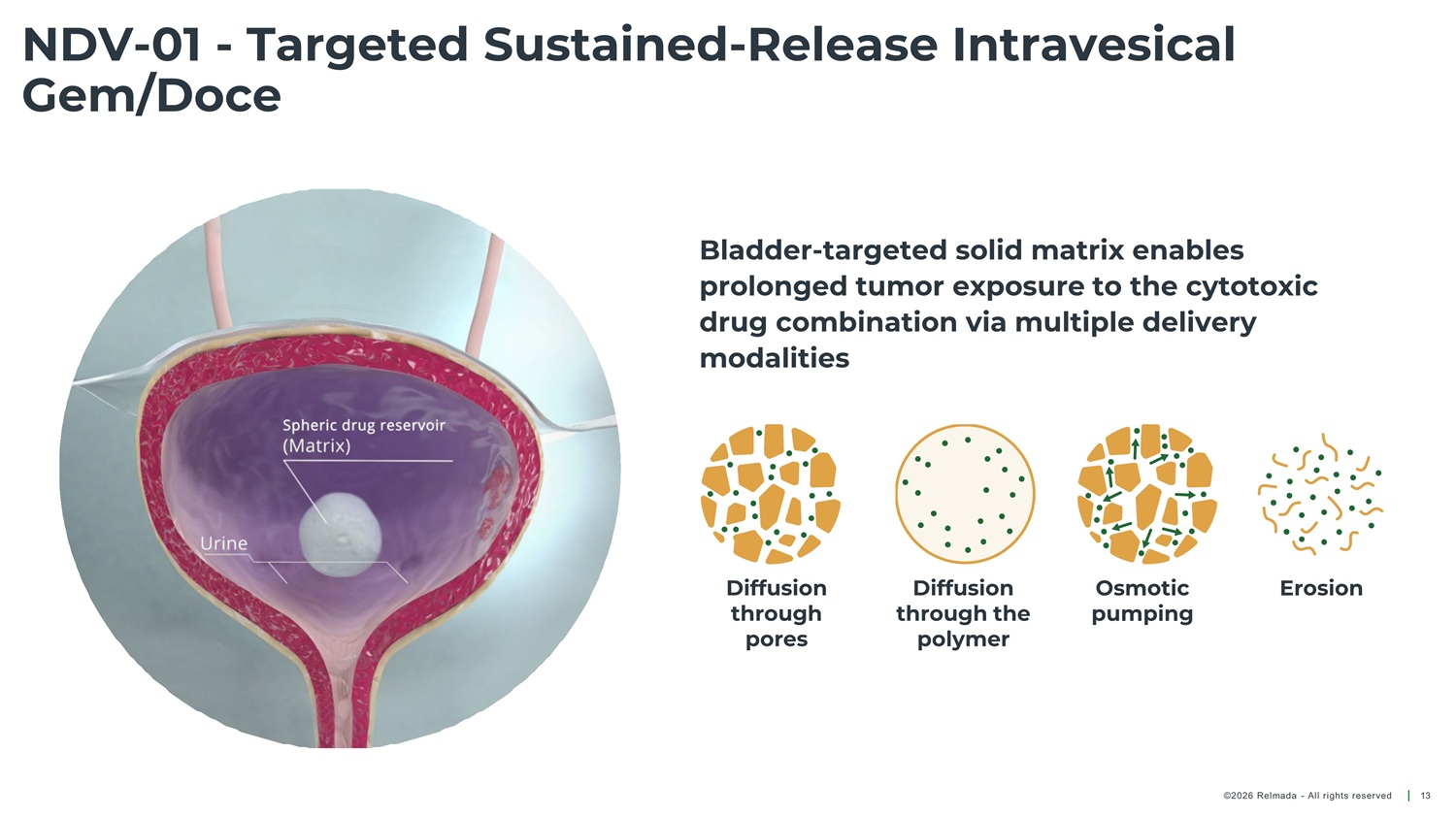
NDV - 01 - Targeted Sustained - Release Intravesical Gem/Doce Bladder - targeted solid matrix enables prolonged tumor exposure to the cytotoxic drug combination via multiple delivery modalities Diffusion through pores Diffusion through the polymer Osmotic pumping Erosion ©2026 Relmada - All rights reserved 13
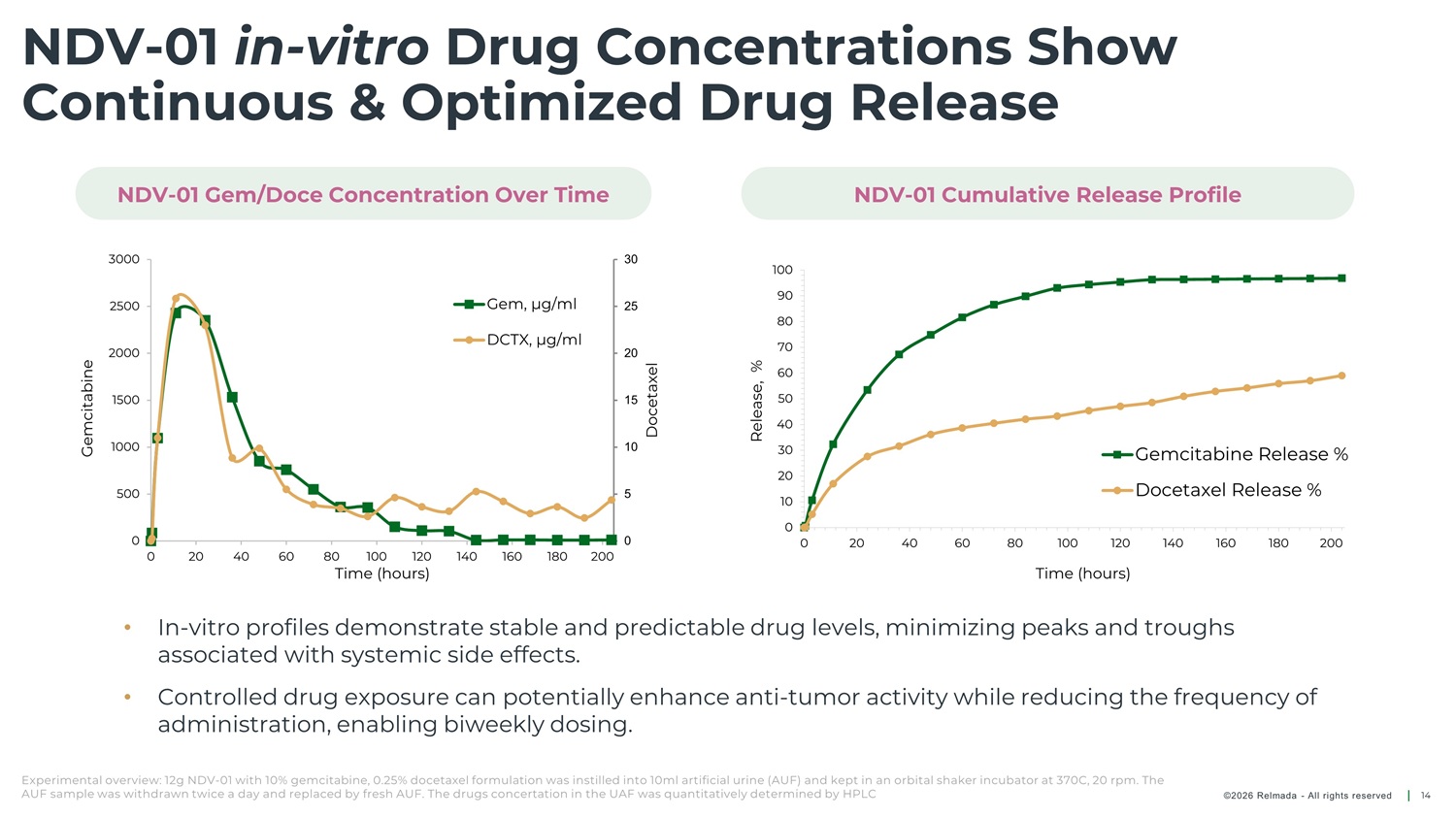
NDV - 01 in - vitro Drug Concentrations Show Continuous & Optimized Drug Release NDV - 01 Gem/Doce Concentration Over Time NDV - 01 Cumulative Release Profile 3000 30 25 20 15 10 5 100 90 80 70 60 50 40 30 20 10 Gem, µg/ml DCTX, µg/ml 2500 2000 1500 1000 500 0 Gemcitabine Release % Docetaxel Release % 0 0 0 20 40 60 80 100 120 140 160 180 200 0 20 40 60 80 100 120 140 160 180 200 Time (hours) Time (hours) • In - vitro profiles demonstrate stable and predictable drug levels, minimizing peaks and troughs associated with systemic side effects. • Controlled drug exposure can potentially enhance anti - tumor activity while reducing the frequency of administration, enabling biweekly dosing. Experimental overview: 12g NDV - 01 with 10% gemcitabine, 0.25% docetaxel formulation was instilled into 10ml artificial urine (AUF) and kept in an orbital shaker incubator at 370C, 20 rpm. The AUF sample was withdrawn twice a day and replaced by fresh AUF. The drugs concertation in the UAF was quantitatively determined by HPLC ©2026 Relmada - All rights reserved 14

NDV - 01: Clinically De - Risked with Clear Competitive Advantages Ready for Use: Rapid, Office - Based Administration NDV - 01 comes as two prefilled syringes instilled in < 5 minutes Convenience: Unlocks Community - Based Treatment In - office administration by MA/RN/LPN without specialized infusion infrastructure, supporting broad adoption in community urology practices where ~80% of NMIBC patients are treated Derisked Based on Conventional Gem/Doce Usage Conventional Gem/Doce is a well - understood and most commonly used in academic practice , providing familiarity and supporting a lower - risk clinical and regulatory pathway Prolonged Intravesical Tumor Exposure NDV - 01 delivers continuous intravesical Gem/Doce inside the bladder enabling sustained tumor exposure Favorable Safety & Clearance Profile The NDV - 01 biodegradable polymer gradually disintegrates and is safely excreted in urine , vs. Inlexz which requires device extraction Relmada internal market research 2025. NMIBC: Non - muscle invasive bladder cancer; Gem/Doce : Gemcitabine plus Docetaxel; MA: Medical assistant; RN: Registered Nurse; LPN: Licensed practical ©2026 Relmada - All rights reserved 15

Study TRCG - 011 for High - Risk NMIBC An open - label, single - arm, single - center Phase 2a study to evaluate safety and efficacy of NDV - 01 in HR NMIBC patients (NCT06663137) HR: High Risk; NMIBC: Non - muscle invasive bladder cancer ©2026 Relmada - All rights reserved 16
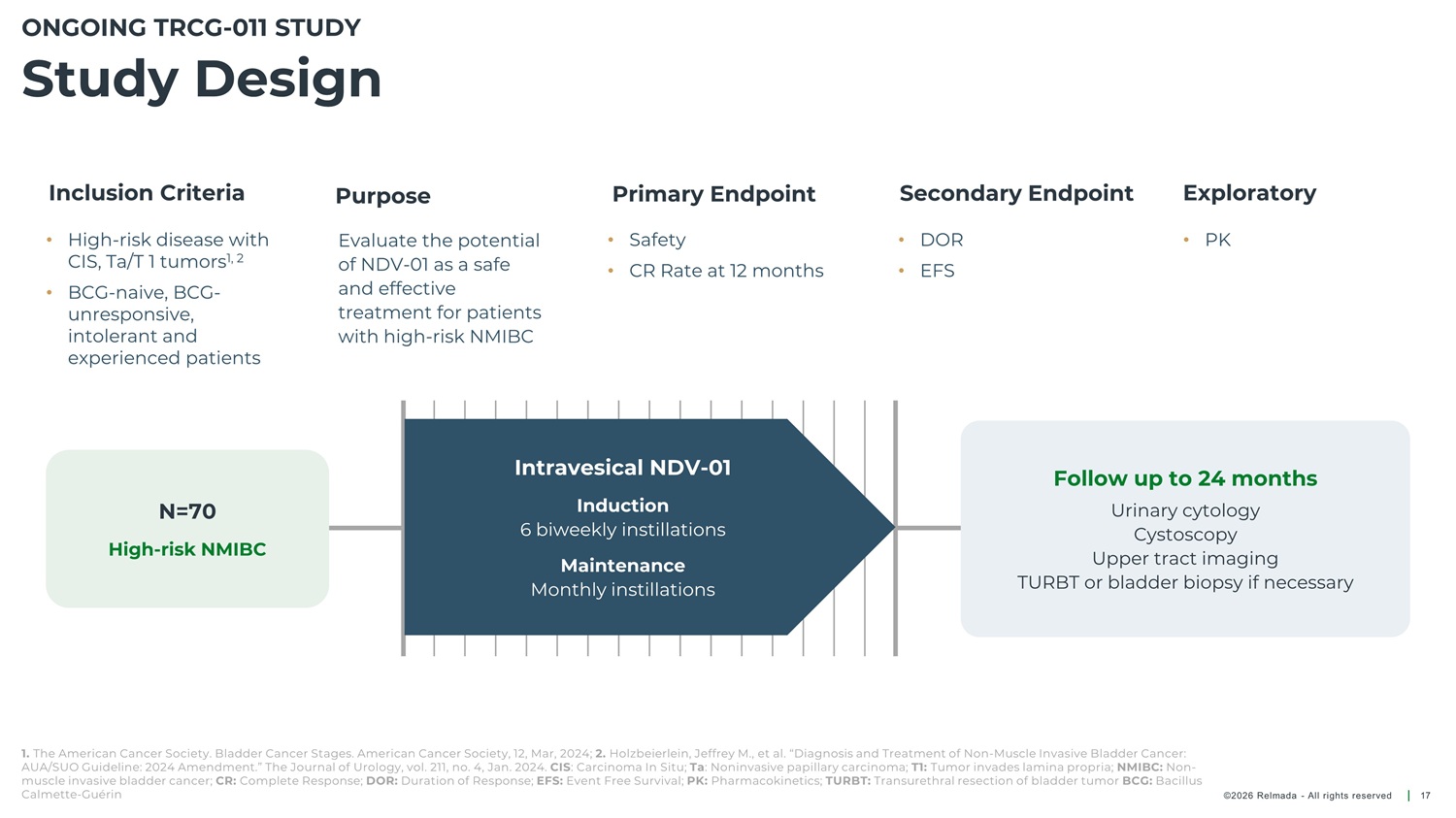
ONGOING TRCG - 011 STUDY Study Design Inclusion Criteria Primary Endpoint Secondary Endpoint Exploratory Purpose • High - risk disease with Evaluate the potential of NDV - 01 as a safe and effective treatment for patients with high - risk NMIBC • Safety • DOR • PK CIS, Ta/T 1 tumors 1, 2 • CR Rate at 12 months • EFS • BCG - naive, BCG - unresponsive, intolerant and experienced patients Intravesical NDV - 01 Follow up to 24 months Induction 6 biweekly instillations N=70 Urinary cytology Cystoscopy Upper tract imaging High - risk NMIBC Maintenance Monthly instillations TURBT or bladder biopsy if necessary 1. The American Cancer Society. Bladder Cancer Stages. American Cancer Society, 12, Mar, 2024; 2. Holzbeierlein, Jeffrey M., et al. “Diagnosis and Treatment of Non - Muscle Invasive Bladder Cancer: AUA/SUO Guideline: 2024 Amendment.” The Journal of Urology, vol. 211, no. 4, Jan. 2024. CIS : Carcinoma In Situ; Ta : Noninvasive papillary carcinoma; T1: Tumor invades lamina propria; NMIBC: Non - muscle invasive bladder cancer; CR: Complete Response; DOR: Duration of Response; EFS: Event Free Survival; PK: Pharmacokinetics; TURBT: Transurethral resection of bladder tumor BCG: Bacillus Calmette - Guérin ©2026 Relmada - All rights reserved 17
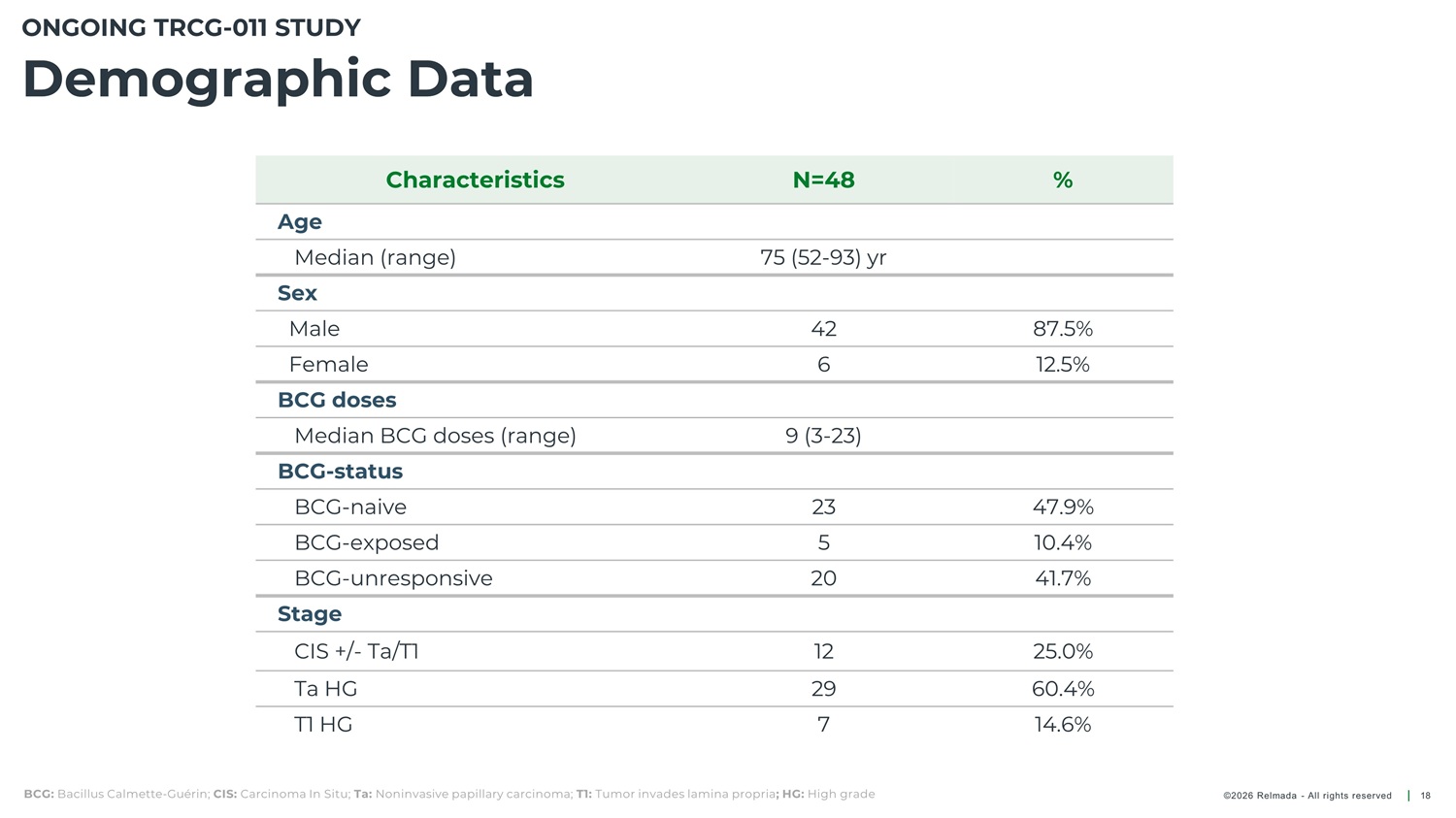
ONGOING TRCG - 011 STUDY Demographic Data Characteristics N=48 % Age Median (range) Sex 75 (52 - 93) yr Male 42 6 87.5% 12.5% Female BCG doses Median BCG doses (range) BCG - status BCG - naive BCG - exposed BCG - unresponsive Stage 9 (3 - 23) 23 5 47.9% 10.4% 41.7% 20 CIS +/ - Ta/T1 Ta HG 12 29 7 25.0% 60.4% 14.6% T1 HG BCG: Bacillus Calmette - Guérin; CIS: Carcinoma In Situ; Ta: Noninvasive papillary carcinoma; T1: Tumor invades lamina propria ; HG: High grade ©2026 Relmada - All rights reserved 18
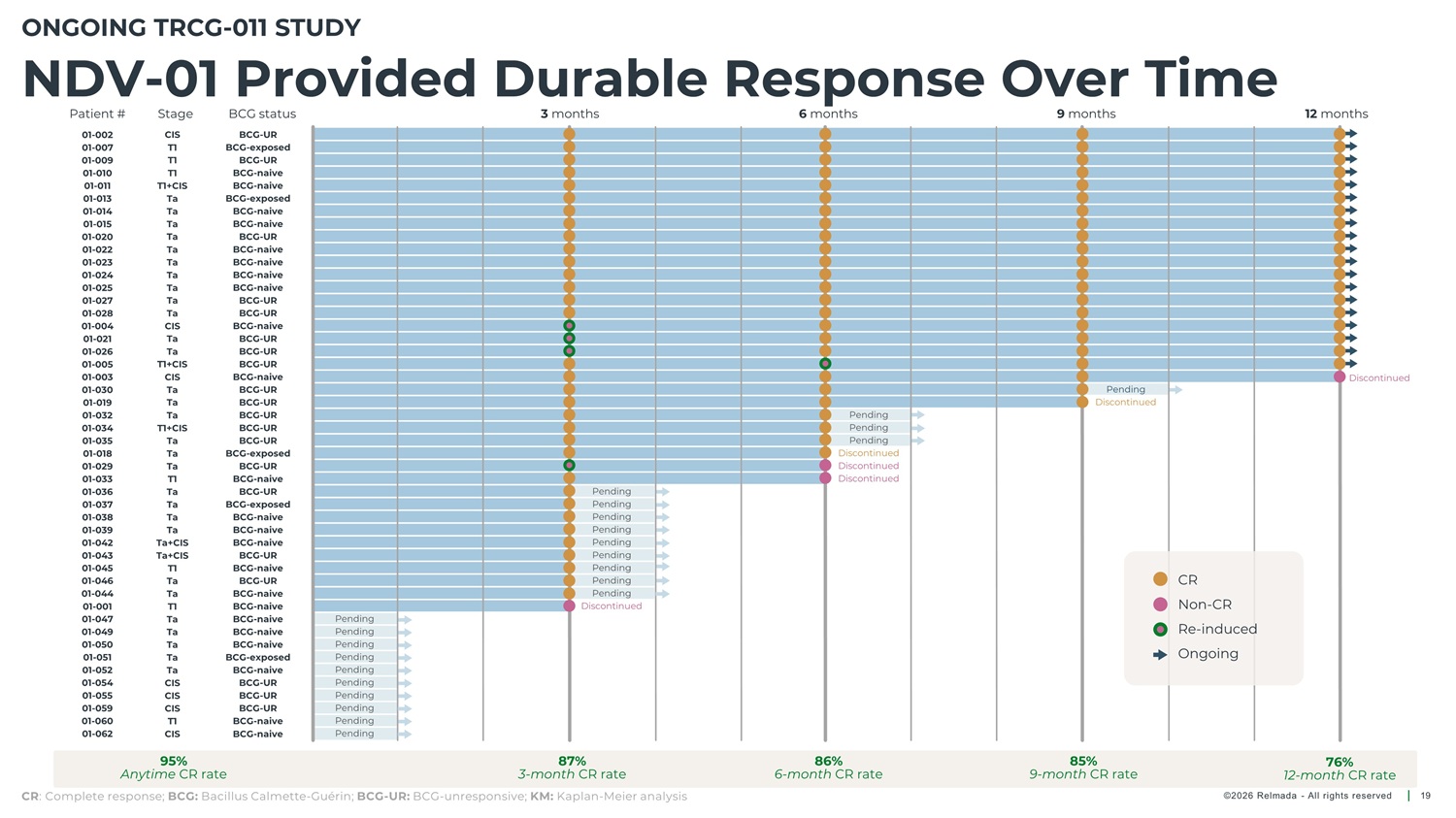
ONGOING TRCG - 011 STUDY NDV - 01 Provided Durable Response Over Time Patient # Stage BCG status 3 months 6 months 9 months 12 months 01 - 002 01 - 007 01 - 009 01 - 010 01 - 011 CIS T1 T1 T1 T1+CIS Ta Ta Ta Ta Ta Ta Ta Ta Ta Ta CIS Ta BCG - UR BCG - exposed BCG - UR BCG - naive BCG - naive BCG - exposed BCG - naive BCG - naive BCG - UR BCG - naive BCG - naive BCG - naive BCG - naive BCG - UR BCG - UR BCG - naive BCG - UR BCG - UR BCG - UR BCG - naive BCG - UR BCG - UR BCG - UR BCG - UR BCG - UR BCG - exposed BCG - UR BCG - naive BCG - UR BCG - exposed BCG - naive BCG - naive BCG - naive BCG - UR 01 - 013 01 - 014 01 - 015 01 - 020 01 - 022 01 - 023 01 - 024 01 - 025 01 - 027 01 - 028 01 - 004 01 - 021 01 - 026 01 - 005 01 - 003 01 - 030 01 - 019 01 - 032 01 - 034 01 - 035 01 - 018 01 - 029 01 - 033 01 - 036 01 - 037 01 - 038 01 - 039 01 - 042 01 - 043 01 - 045 01 - 046 01 - 044 01 - 001 01 - 047 01 - 049 01 - 050 01 - 051 01 - 052 01 - 054 01 - 055 01 - 059 01 - 060 01 - 062 Ta T1+CIS CIS Ta Ta Ta T1+CIS Ta Ta Ta T1 Ta Ta Ta Ta Ta+CIS Ta+CIS T1 Ta Ta T1 Ta Ta Ta Ta Ta CIS CIS CIS T1 Discontinued Pending Discontinued Pending Pending Pending Discontinued Discontinued Discontinued Pending Pending Pending Pending Pending Pending Pending Pending Pending Discontinued BCG - naive BCG - UR CR BCG - naive BCG - naive BCG - naive BCG - naive BCG - naive BCG - exposed BCG - naive BCG - UR BCG - UR BCG - UR BCG - naive BCG - naive Non - CR Re - induced Ongoing Pending Pending Pending Pending Pending Pending Pending Pending Pending Pending CIS 95% 87% 3 - month CR rate 86% 6 - month CR rate 85% 9 - month CR rate 76% 12 - month CR rate Anytime CR rate CR : Complete response; BCG: Bacillus Calmette - Guérin; BCG - UR: BCG - unresponsive; KM: Kaplan - Meier analysis ©2026 Relmada - All rights reserved 19
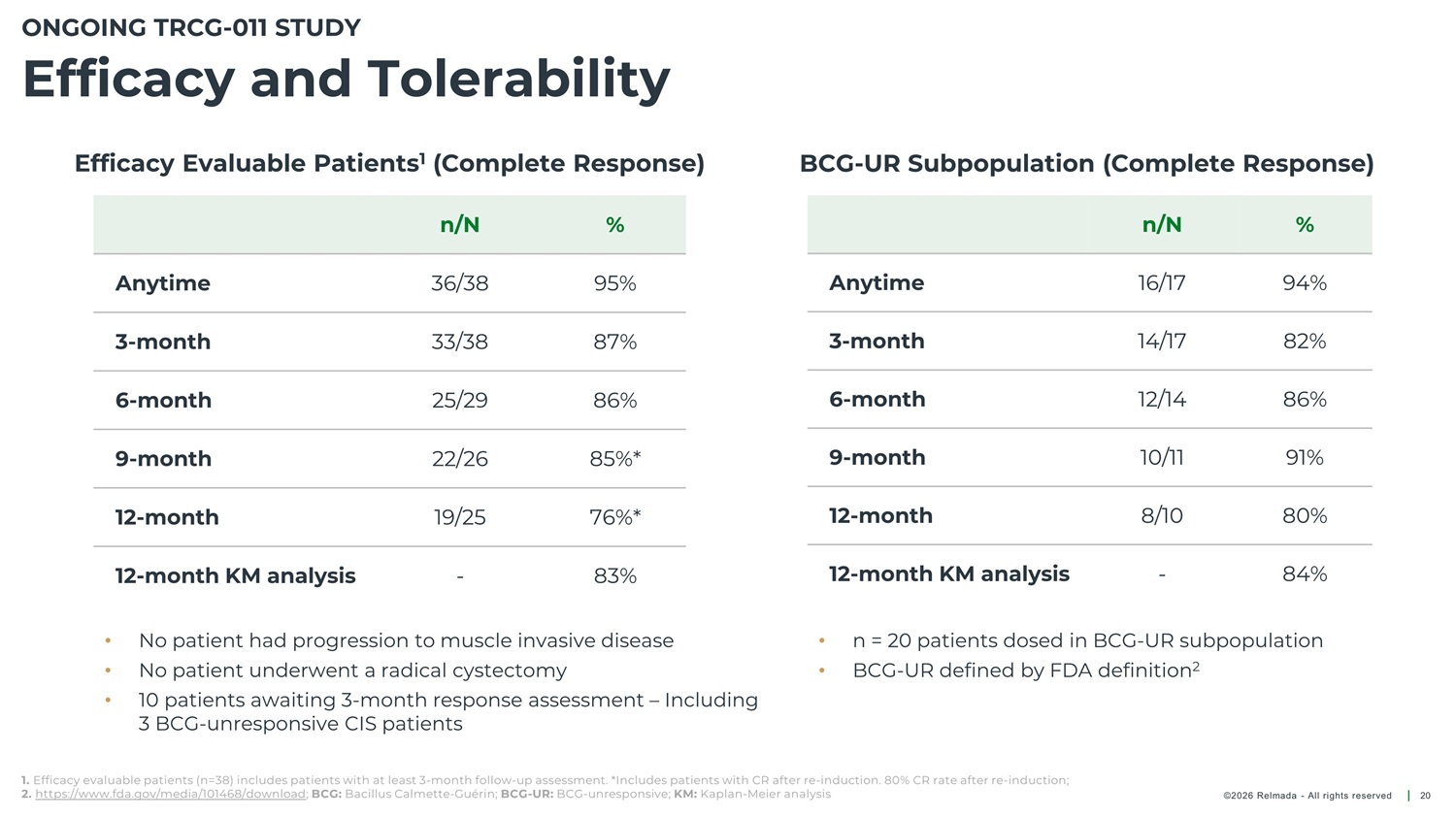
ONGOING TRCG - 011 STUDY Efficacy and Tolerability Efficacy Evaluable Patients 1 (Complete Response) BCG - UR Subpopulation (Complete Response) n/N 36/38 33/38 25/29 22/26 19/25 - % n/N 16/17 14/17 12/14 10/11 8/10 - % Anytime 95% 87% 86% 85%* 76%* 83% Anytime 94% 82% 86% 91% 80% 84% 3 - month 3 - month 6 - month 6 - month 9 - month 9 - month 12 - month 12 - month 12 - month KM analysis 12 - month KM analysis • • • No patient had progression to muscle invasive disease No patient underwent a radical cystectomy 10 patients awaiting 3 - month response assessment – Including 3 BCG - unresponsive CIS patients • • n = 20 patients dosed in BCG - UR subpopulation BCG - UR defined by FDA definition 2 1. Efficacy evaluable patients (n=38) includes patients with at least 3 - month follow - up assessment. *Includes patients with CR after re - induction. 80% CR rate after re - induction; 2. https://www.fda.gov/media/101468/download ; BCG: Bacillus Calmette - Guérin; BCG - UR: BCG - unresponsive; KM: Kaplan - Meier analysis ©2026 Relmada - All rights reserved 20

ONGOING TRCG - 011 STUDY Treatment - Related AE and Tolerability No patient had ≥ Grade 3 TRAE No patients discontinued treatment due to AEs Of the 48 patients who received ≥ 1 dose of NDV - 01, 30 (63%) had a TRAE • 54% transient uncomfortable urination (dysuria) • 8% asymptomatic positive urine culture • 8% hematuria TRAE: Treatment - related adverse events; AE: Adverse events ©2026 Relmada - All rights reserved 21
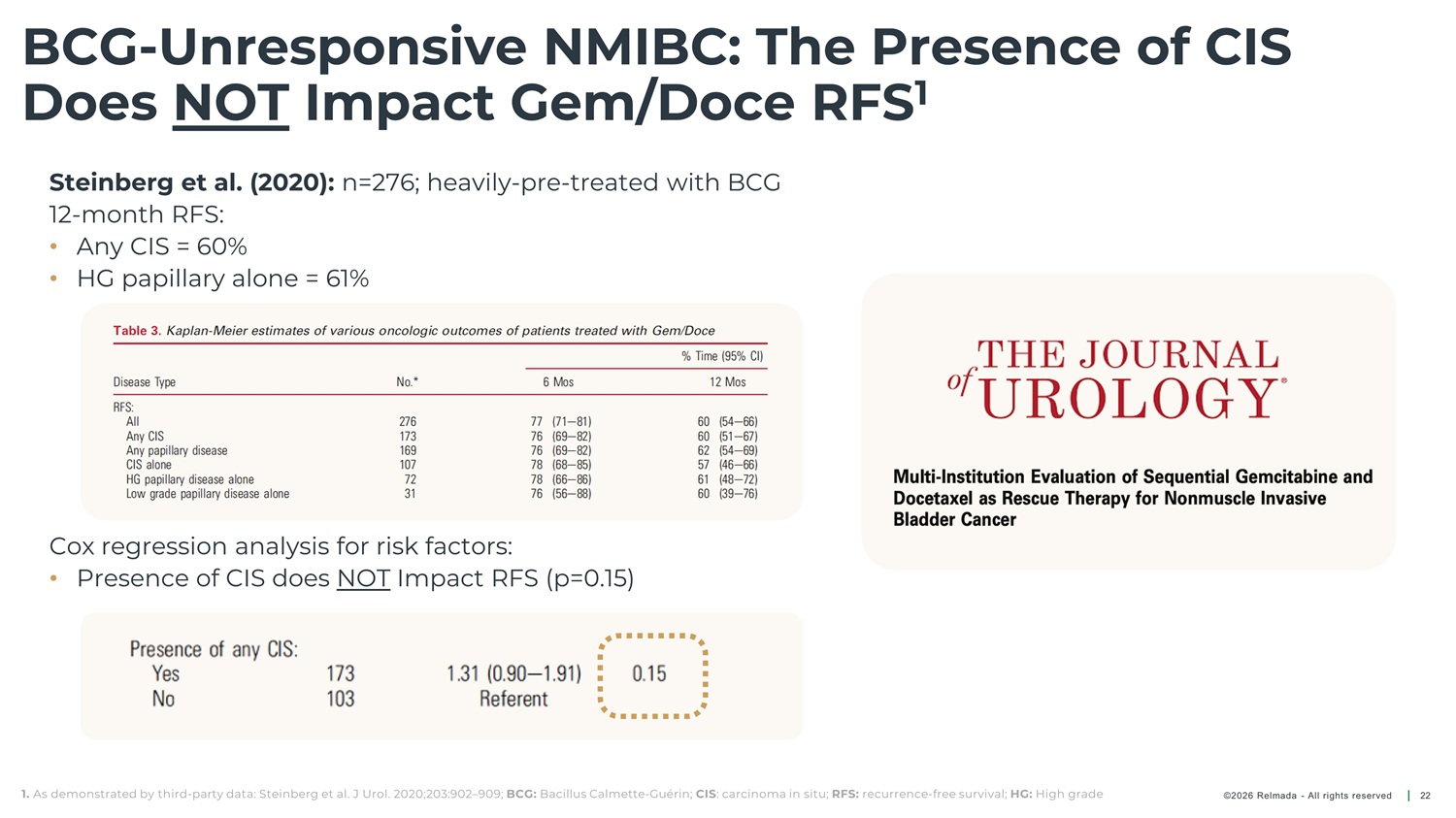
BCG - Unresponsive NMIBC: The Presence of CIS Does NOT Impact Gem/Doce RFS 1 Steinberg et al. (2020): n=276; heavily - pre - treated with BCG 12 - month RFS: • Any CIS = 60% • HG papillary alone = 61% Cox regression analysis for risk factors: • Presence of CIS does NOT Impact RFS (p=0.15) 1. As demonstrated by third - party data: Steinberg et al. J Urol. 2020;203:902 – 909; BCG: Bacillus Calmette - Guérin; CIS : carcinoma in situ; RFS: recurrence - free survival; HG: High grade ©2026 Relmada - All rights reserved 22

Phase 3 Program R ecurrent / E ndovesical / S urgery - sparing / C ombination therapy for / U rothelial cancer / E ffectiveness ©2026 Relmada - All rights reserved 23

PHASE 3 RESCUE TRIAL Two Independent NDV - 01 Approval Pathways Provide Significant Market Opportunity Registrational Pathway 2 Registrational Pathway 1 Open label randomized controlled trial in intermediate - risk NMIBC – adjuvant therapy following TURBT (NDV - 01 vs. observation) Single - arm trial in 2L BCG - unresponsive NMIBC with CIS who are refractory to approved or developmental therapies ~75k patients/annually in US 1 – with ~35% 2 of intermediate - risk patients receiving adjuvant therapy post - TURBT ~5k patients/annually in US 1 – based on 12 - month CR rates of 19% - 46% 3 for 1L BCG - unresponsive therapies 1. Based on Internal estimates. 2. Grabe - Heyne et al. Front Oncol. 2023. 3. FDA approval summaries; company disclosures; published clinical trial data. NMIBC: Non - muscle invasive bladder cancer; BCG: Bacillus Calmette - Guérin (BCG); TURBT: Transurethral Resection of Bladder Tumor; CIS : carcinoma in situ; 1L: first - line; 2L: second - line; CR : Complete Response; ©2026 Relmada - All rights reserved 24

PHASE 3 RESCUE TRIAL Cohort 2A: 2L BCG - Unresponsive NMIBC Open - label, single - arm study to evaluate safety and efficacy of NDV - 01 in BCG - UR refractory to first - line therapy Inclusion Criteria Purpose Primary Endpoint Secondary Endpoint Other • HR BCG - UR with CIS refractory to first - line therapy • Safety and efficacy of NDV - 01 in patients with HR BCG - UR with CIS • CR anytime • Safety • DOR • PK • PFS • RFS amongst responders Study design Intravesical NDV - 01 Follow up to 24 months Cohort 2A 1 (Registrational; N=87) Induction 6 biweekly instillations Urinary cytology Cystoscopy TURBT or bladder biopsy if necessary HR BCG - UR NMIBC with CIS refractory to 1 st line therapy Maintenance Monthly instillations 1. BCG - Unresponsive patients with CIS +/ - Ta/T1 disease. Phase 3 Cohort 2A is a registrational cohort intended for regulatory approval. 2. BCG - Unresponsive patients with high - grade Ta/T1 disease. Phase 2 Cohort 2B is an exploratory cohort and not intended for regulatory approval. HR: High risk; CIS: Carcinoma In Situ; CR: Complete Response; DOR: Duration of Response; RFS: Recurrence Free Survival; PFS: Progression Free Survival; PK: Pharmacokinetics; TURBT: Transurethral resection of bladder tumor; BCG: Bacillus Calmette - Guérin BCG - UR: BCG - unresponsive ©2026 Relmada - All rights reserved 25
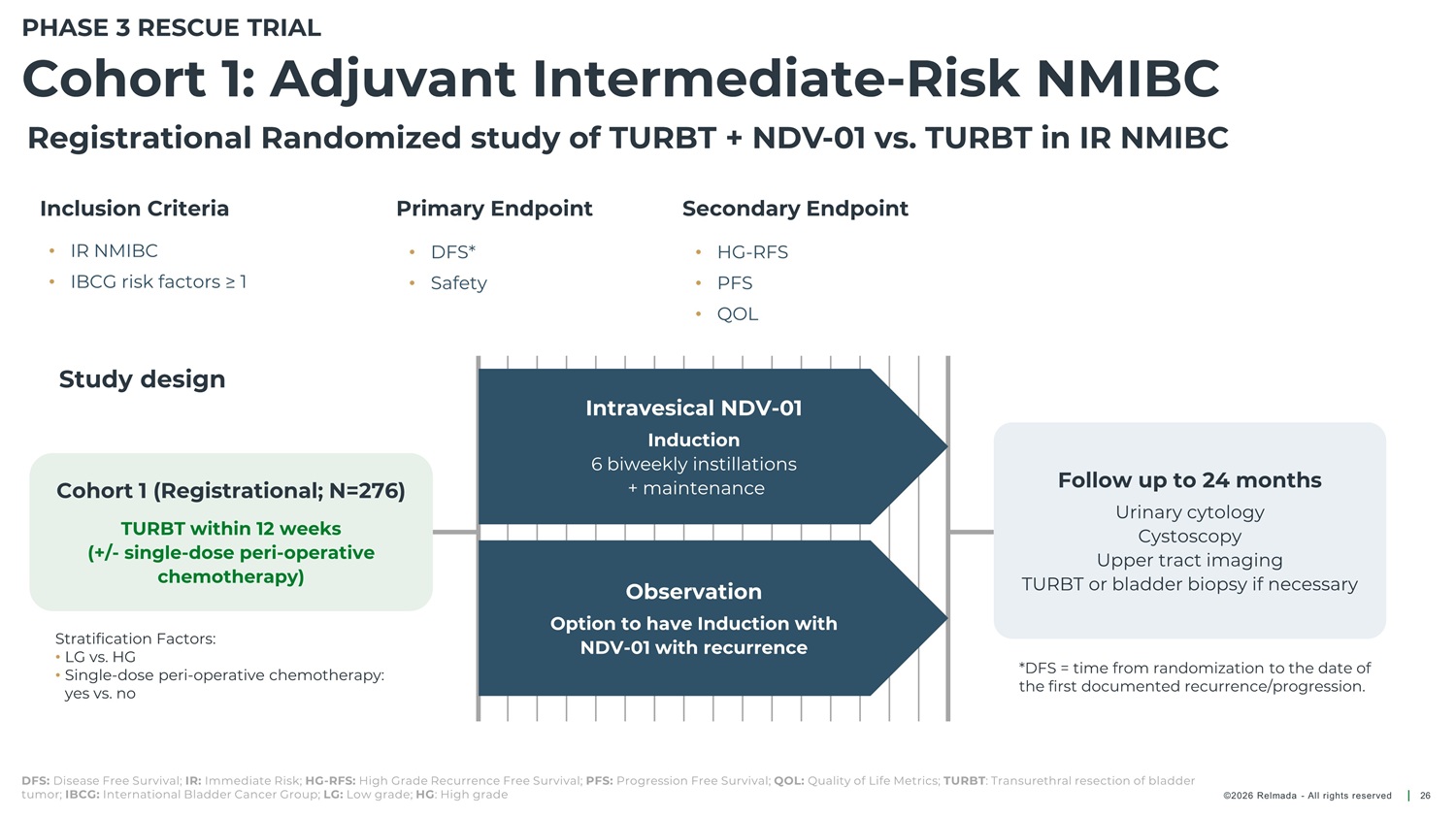
PHASE 3 RESCUE TRIAL Cohort 1: Adjuvant Intermediate - Risk NMIBC Registrational Randomized study of TURBT + NDV - 01 vs. TURBT in IR NMIBC Inclusion Criteria Primary Endpoint Secondary Endpoint • IR NMIBC DFS* • HG - RFS • PFS • • • IBCG risk factors ≥ 1 Safety • QOL Study design Intravesical NDV - 01 Induction 6 biweekly instillations + maintenance Follow up to 24 months Cohort 1 (Registrational; N=276) Urinary cytology Cystoscopy Upper tract imaging TURBT within 12 weeks (+/ - single - dose peri - operative chemotherapy) TURBT or bladder biopsy if necessary Observation Option to have Induction with NDV - 01 with recurrence Stratification Factors: • LG vs. HG • Single - dose peri - operative chemotherapy: yes vs. no *DFS = time from randomization to the date of the first documented recurrence/progression. DFS: Disease Free Survival; IR: Immediate Risk; HG - RFS: High Grade Recurrence Free Survival; PFS: Progression Free Survival; QOL: Quality of Life Metrics; TURBT : Transurethral resection of bladder tumor; IBCG: International Bladder Cancer Group; LG: Low grade; HG : High grade ©2026 Relmada - All rights reserved 26

Expecting to Advance NDV - 01 Towards Registration - Track Studies in Mid - 2026 Initiate Phase 3 RESCUE Trials Target two independent registrational pathways: • 2L BCG - Unresponsive NMIBC patients Mid 2026 • Adjuvant Intermediate - Risk NMIBC patients Interim Phase 3 2L BCG - Unresponsive 3 - month Data Initial 3 - month CR data + safety Q4 2026 BCG: Bacillus Calmette - Guérin (BCG); NMIBC: No muscle invasive bladder cancer; IR: Intermediate Risk ©2026 Relmada - All rights reserved 27

Sepranolone A novel candidate, with potential to overcome the challenges of current therapies for compulsivity disorders ©2026 Relmada - All rights reserved 28
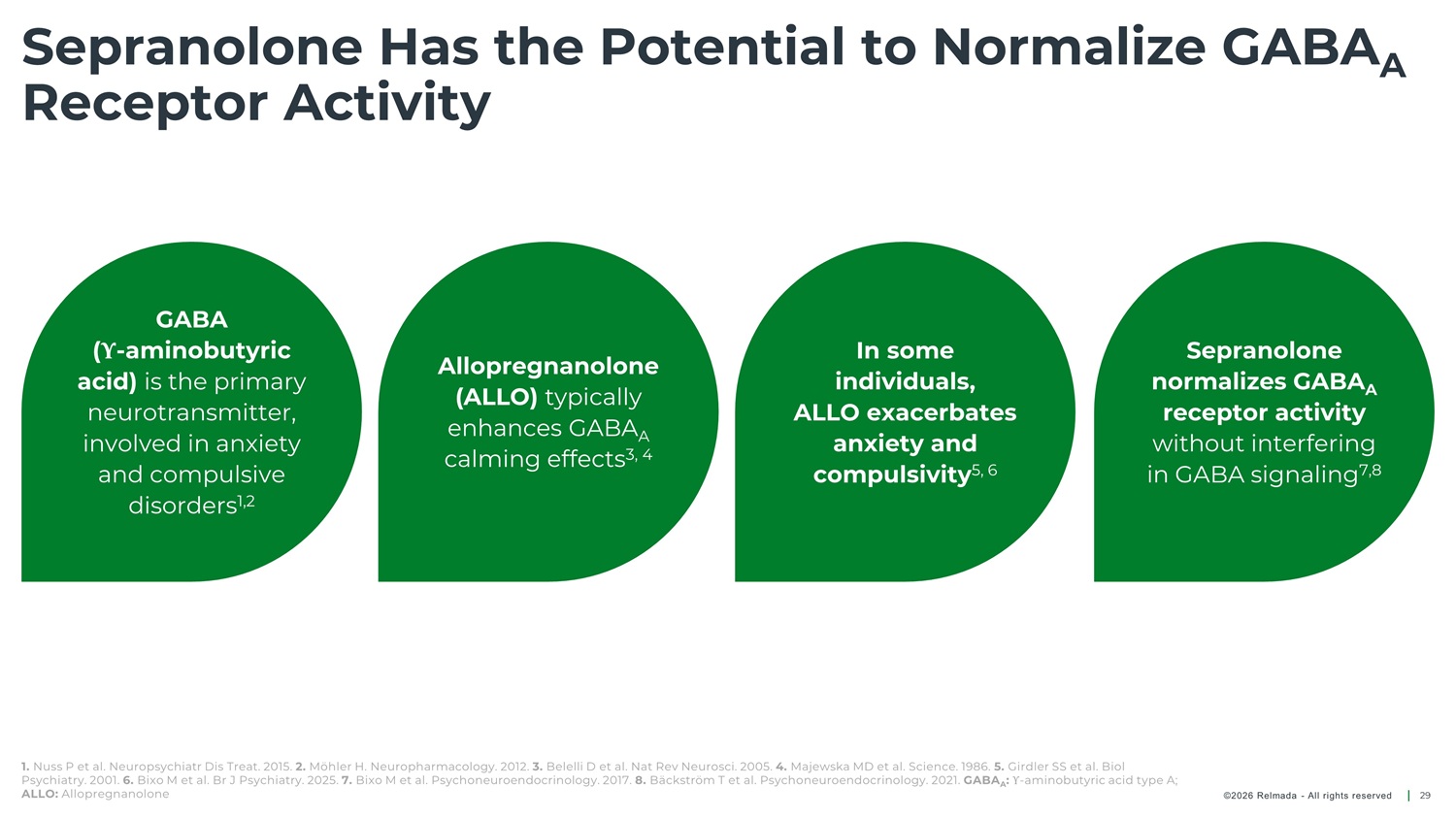
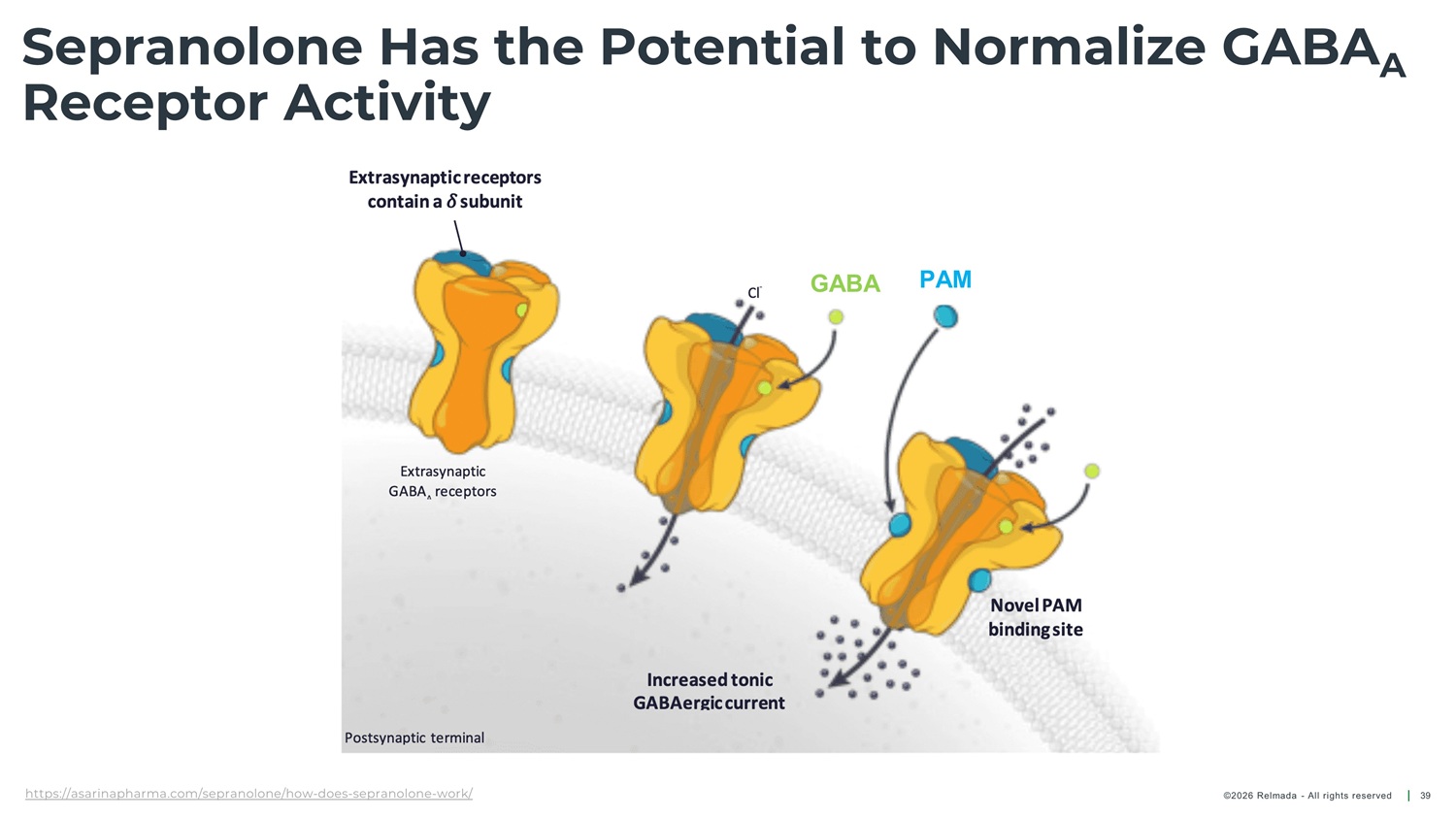
Sepranolone Has the Potential to Normalize GABA A Receptor Activity GABA ( Υ - aminobutyric acid) is the primary neurotransmitter, involved in anxiety and compulsive disorders 1,2 In some individuals, ALLO exacerbates anxiety and Sepranolone Allopregnanolone normalizes GABA A receptor activity without interfering in GABA signaling 7,8 (ALLO) typically enhances GABA calming effects 3, 4 A compulsivity 5, 6 1. Nuss P et al. Neuropsychiatr Dis Treat. 2015. 2. Möhler H. Neuropharmacology. 2012. 3. Belelli D et al. Nat Rev Neurosci. 2005. 4. Majewska MD et al. Science. 1986. 5. Girdler SS et al. Biol Psychiatry. 2001. 6. Bixo M et al. Br J Psychiatry. 2025. 7. Bixo M et al. Psychoneuroendocrinology. 2017. 8. Bäckström T et al. Psychoneuroendocrinology. 2021. GABA : Υ - aminobutyric acid type A; A ALLO: Allopregnanolone ©2026 Relmada - All rights reserved 29
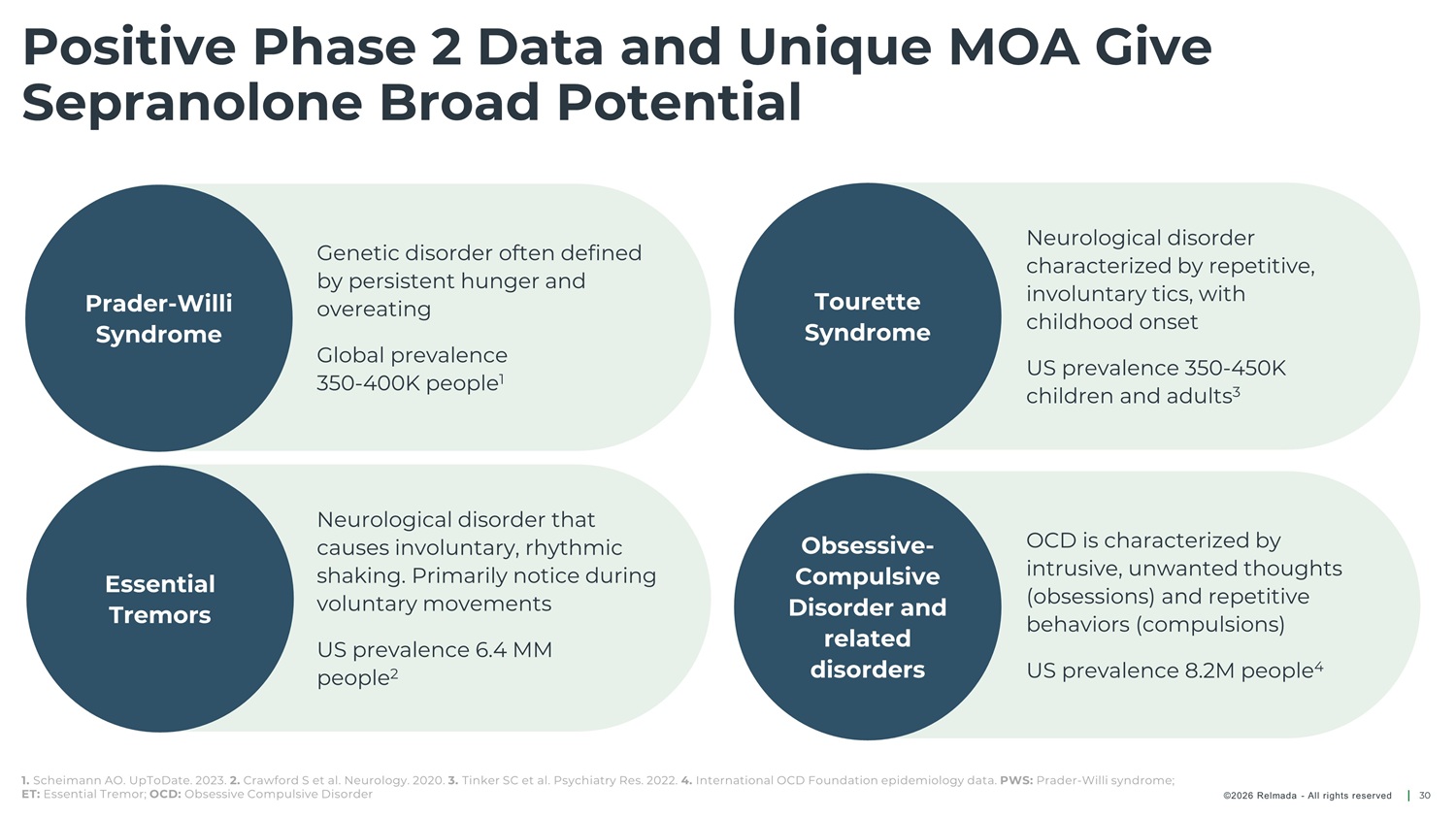
Positive Phase 2 Data and Unique MOA Give Sepranolone Broad Potential Neurological disorder characterized by repetitive, involuntary tics, with childhood onset Genetic disorder often defined by persistent hunger and overeating Prader - Willi Syndrome Tourette Syndrome Global prevalence 350 - 400K people 1 US prevalence 350 - 450K children and adults 3 Neurological disorder that causes involuntary, rhythmic shaking. Primarily notice during voluntary movements OCD is characterized by intrusive, unwanted thoughts (obsessions) and repetitive behaviors (compulsions) Obsessive - Compulsive Disorder and related Essential Tremors US prevalence 6.4 MM people 2 disorders US prevalence 8.2M people 4 1. Scheimann AO. UpToDate. 2023. 2. Crawford S et al. Neurology. 2020. 3. Tinker SC et al. Psychiatry Res. 2022. 4. International OCD Foundation epidemiology data. PWS: Prader - Willi syndrome; ET: Essential Tremor; OCD: Obsessive Compulsive Disorder ©2026 Relmada - All rights reserved 30

Sepranolone: Highlights & Development Value Differentiated therapeutic candidate for compulsivity - related disorders, supported by positive proof - of - concept data in Tourette’s syndrome Phase 2 study in Prader - Willi syndrome (PWS) planned for H1 2026, targeting a rare genetic disorder affecting 350,000 – 400,000 individuals worldwide Program readiness: Regulatory engagement and manufacturing activities are actively underway, supporting efficient trial initiation Orphan/rare disease incentives: Potential for orphan drug designation, including regulatory exclusivity, accelerated approval pathways, and enhanced commercial visibility Strategic investor value: Clear development milestones, potential for first - in - class differentiation, and meaningful opportunity in a high - unmet - need rare disease ©2026 Relmada - All rights reserved 31

Expecting to Advance Sepranolone Towards Phase 2 Study in Prader - Willi Syndrome in Mid - 2026 Mid 2026 Initiation of Pilot Phase 2 study in Prader - Willi Syndrome Focus on evaluating early proof - of - concept PWS: Prader - Willi syndrome ©2026 Relmada - All rights reserved 32

Corporate Summary ©2026 Relmada - All rights reserved 33
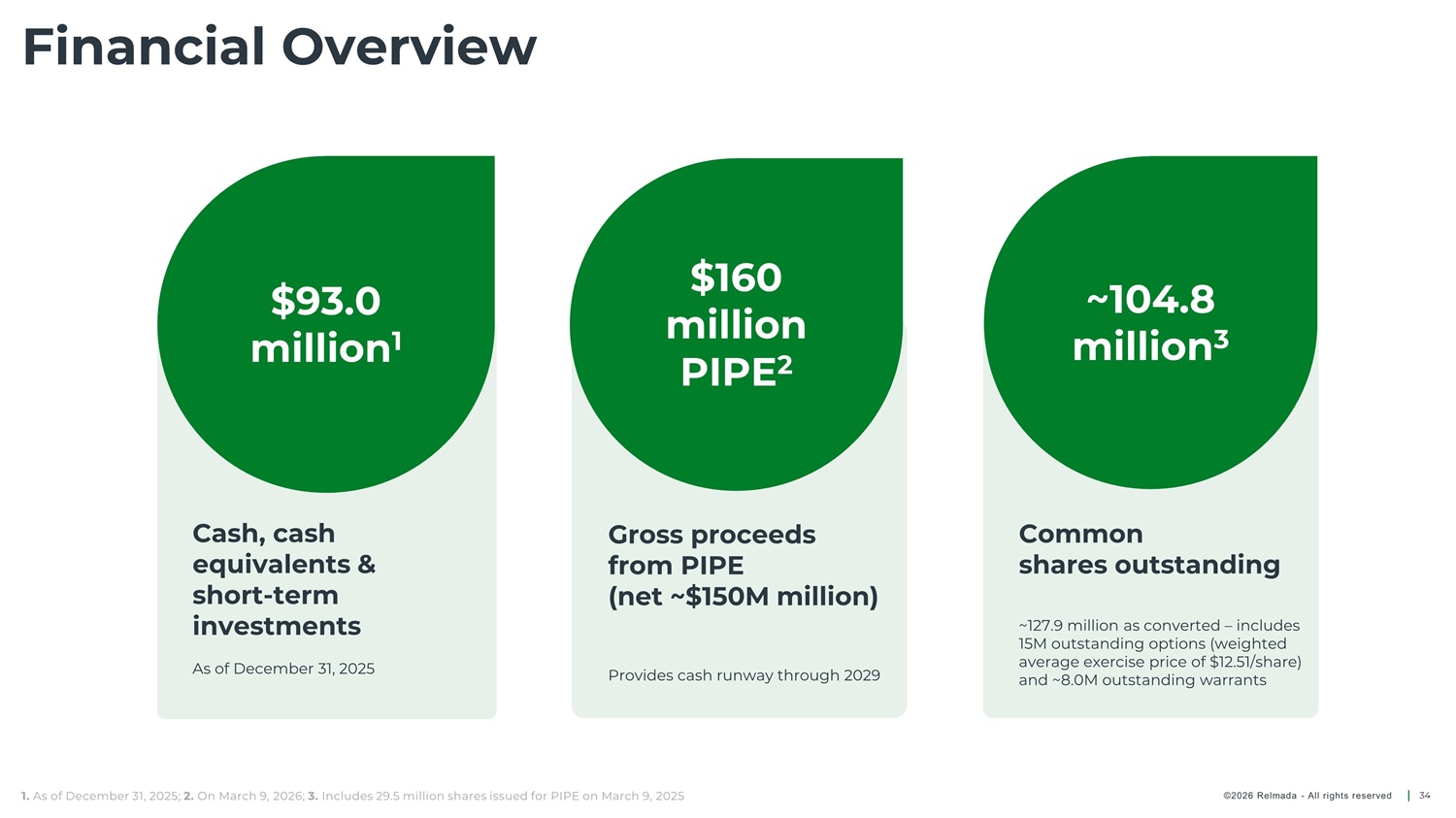
Financial Overview $160 million PIPE 2 $93.0 million ~104.8 million 3 1 Cash, cash Gross proceeds from PIPE Common equivalents & short - term investments shares outstanding (net ~$150M million) ~127.9 million as converted – includes 15M outstanding options (weighted average exercise price of $12.51/share) and ~8.0M outstanding warrants As of December 31, 2025 Provides cash runway through 2029 1. As of December 31, 2025; 2. On March 9, 2026; 3. Includes 29.5 million shares issued for PIPE on March 9, 2025 34 ©2026 Relmada - All rights reserved

Thank You!

Appendix

Gem/Doce combination has been embraced by the urologic oncology community Effective salvage treatment for patients who have failed or are intolerant to BCG with reported 2 - year RFS ~50% 1, 2, 3 Gem/Doce is an effective alternative first - line agent in high - risk BCG naïve patients with 2 - year RFS of 82% 4 Gem/Doce use expanding into intermediate - risk and low - grade tumors with reported 2 - year RFS of 70 - 80% 5, 6 Gem/Doce avoids/delays radical cystectomy 7, 8 Large ongoing cooperative “BRIDGE” study (n=870) evaluating Gem/Doce combination vs. BCG (NCT05538663) 1. Steinberg RL et al. J Urol. 2020; 2. Garneau CA et al. Can Urol Assoc J. 2024; 3. Yim K et al. Urol Oncol. 2023; 4. McElree IM et al. J Urol. 2022; 5. McElree IM et al. Urol Oncol. 2023; 6. Tan WS et al. Eur Urol Oncol. 2023; 7. Chevuru PT et al. Urol Oncol. 2023; 8. Narayan VM et al. J Urol. 2024. 9 . Steinberg RL et al. J Urol. 2019; RFS: Relapse Free Survival; BCG: Bacillus Calmette - Guérin; NMIBC: Non - muscle invasive bladder cancer; Gem/Doce : Gemcitabine plus Docetaxel ©2026 Relmada - All rights reserved 37

PHASE 3 RESCUE TRIAL Cohort 2B: 2L BCG - Unresponsive NMIBC Open - label, single - arm study to evaluate safety and efficacy of NDV - 01 in BCG - UR refractory to first - line therapy Inclusion Criteria Purpose Primary Endpoint Secondary Endpoint Other • HR BCG - UR papillary only refractory to • Safety and efficacy of NDV - 01 in patients with HR BCG - UR with CIS • CR anytime • Safety • DOR • PK • PFS first - line therapy • RFS amongst responders Study design Intravesical NDV - 01 Follow up to 24 months Cohort 2B 1 (Exploratory; N=30) Induction 6 biweekly instillations Urinary cytology Cystoscopy HR BCG - UR NMIBC papillary only refractory to 1 st line therapy TURBT or bladder biopsy if necessary Maintenance Monthly instillations 1. BCG - Unresponsive patients with high - grade Ta/T1 disease. Phase 2 Cohort 2B is an exploratory cohort and not intended for regulatory approval. CR: Complete Response; DOR: Duration of Response; RFS: Recurrence Free Survival; PFS: Progression Free Survival; BCG - UR: BCG - unresponsive ©2026 Relmada - All rights reserved 38
Sepranolone Has the Potential to Normalize GABA A Receptor Activity PAM GABA https://asarinapharma.com/sepranolone/how - does - sepranolone - work/ ©2026 Relmada - All rights reserved 39

Management Leadership Board of Directors Sergio Traversa Maged Shenouda Chuck Ence Chief Accounting and Charles J. Casamento John Glasspool Fabiana Fedeli Chief Executive Officer Chief Financial Officer Chairman of the Board Director Director Compliance Officer Paul Kelly Chief Operating Officer Raj S. Pruthi, MD Chief Medical Officer Sergio Traversa Chief Executive Officer Paul Kel ly Chief Operating Officer ©2026 Relmada - All rights reserved 40